The Arthur E. Schwarting Symposium is an educational conference focused on pharmacy practice for pharmacists in many settings.
This year's sympoisum had an overall topic of Drug Induced Disease from a Patient Safety perspective. This presentation deals with drug induced cardiovascular issues.
Learning Objectives
The activity met the following learning objectives for Pharmacists:
| · Identify heart block |
| · Differentiate between 1st, type-1 2nd, type-2 2nd, and 3rd degree AV block |
| · Describe the drugs and dietary supplements that can cause heard block and what to do if drug induced heart block occurs acutely and chronically |
| · Identify QTc interval prolongation and describe how much of an elevation dramatically enhances the risk of Torsade de Pointes |
| · Describe drugs and dietary supplements that may prolong QTc interval and interventions for acute Torsade de Pointes or chronic QTc interval prolongation |
| · Apply knowledge to a patient case |
Session Offered
Released: May 13, 2021
Expires: May 13, 2024
Course Fee
$15 Pharmacist
ACPE UAN Codes
0009-0000-21-007-H05-P
Session Code
21RW07-ABC37
Accreditation Hours
1.0 hours of CE
Accreditation Statement
| The University of Connecticut School of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education. |  |
Pharmacists and Pharmacy Technicians are eligible to participate in this application-based activity and will receive 1.0 CE Hour for completing the activity (ACPE UAN 0009-0000-21-007-H05-P), passing the quiz with a grade of 70% or better, and completing an online evaluation. Statements of credit are available via the CPE Monitor online system and your participation will be recorded with CPE Monitor within 72 hours of submission.
Grant Funding
There is no grant funding for this activity.
Faculty
Michael White, PharmD, FCCP, FCP
BOT Distinguished Professor and Chair of Pharmacy Practice
University of Connecticut School of Pharmacy
Storrs, CT
Faculty Disclosure
Dr. White has no financial relationships with any ineligible company associated with these presentations.
Disclaimer
The material presented here does not necessarily reflect the views of The University of Connecticut School of Pharmacy or its co-sponsor affiliates. These materials may discuss uses and dosages for therapeutic products, processes, procedures and inferred diagnoses that have not been approved by the United States Food and Drug Administration. A qualified health care professional should be consulted before using any therapeutic product discussed. All readers and continuing education participants should verify all information and data before treating patients or employing any therapies described in this continuing education activity.
Content
Post Test Pharmacist
Patient Safety: Drug Induced Cardiovascular Disease
At the conclusion of this lecture the successful learner will be able to:
- Identify heart block
- Differentiate between 1st, type-1 2nd, type-2 2nd, and 3rd degree AV block
- Describe the drugs and dietary supplements that can cause heart block and what to do if drug induced heart block occurs acutely and chronically
- Identify QTc interval prolongation and describe how much of an elevation dramatically enhances the risk of Torsade de Pointes
- Describe drugs and dietary supplements that may prolong QTc interval and interventions for acute Torsade de Pointes or chronic QTc interval prolongation
- Apply knowledge to a patient relevant case
1. Using the rhythm strip below, what is the PP interval and the RR interval in seconds?
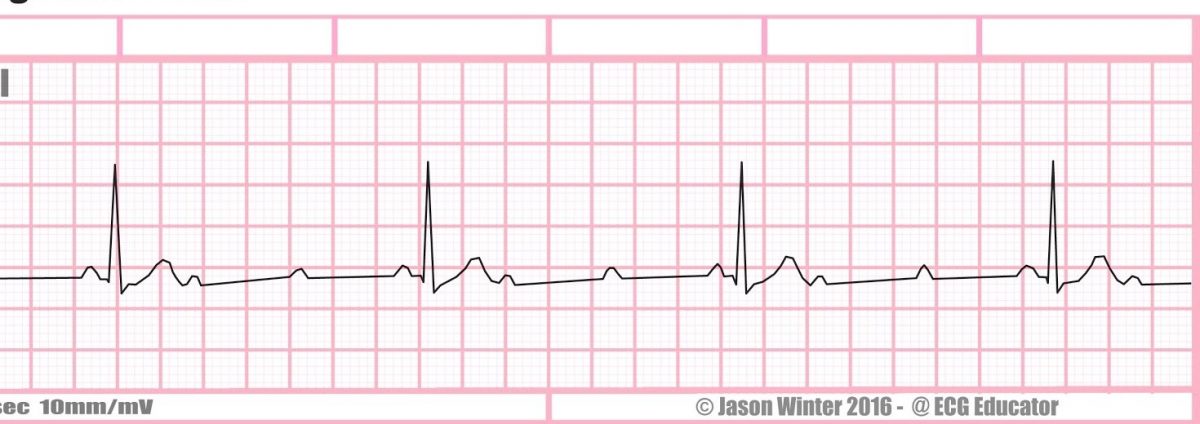
a. PP interval = 0.52 s, RR interval = 1.44 s
b. PP interval = 1.44 s, RR interval = 0.52 s
c. PP interval = 0.73 s, RR interval = 0.73 s
2. What arrhythmia is shown on this rhythm strip?
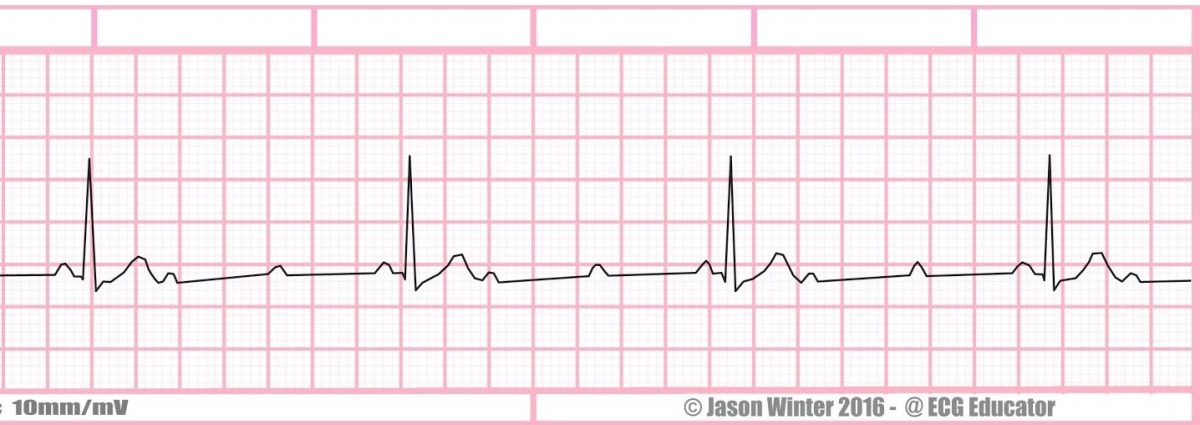
a. First degree AV block
b. Third degree AV block
c. Torsade de Pointes
3. Which of the following rhythm issues is unlikely to be induced by a drug that blocks the AV node?
a. First degree AV block
b. Type I second degree AV block
c. Type II second degree AV block
4. Which of the following drugs is least likely to cause drug induced AV block
a. Amlodipine
b. Metoprolol
c. Digoxin
5. If a patient has symptomatic heart block due to the use of drugs that can induce heart block, which of the following can be used to treat it?
a. Gabapentin
b. Propranolol
c. Atropine
6. Using the rhythm strip below, what is the QT interval, RR interval, and QTc interval in seconds?
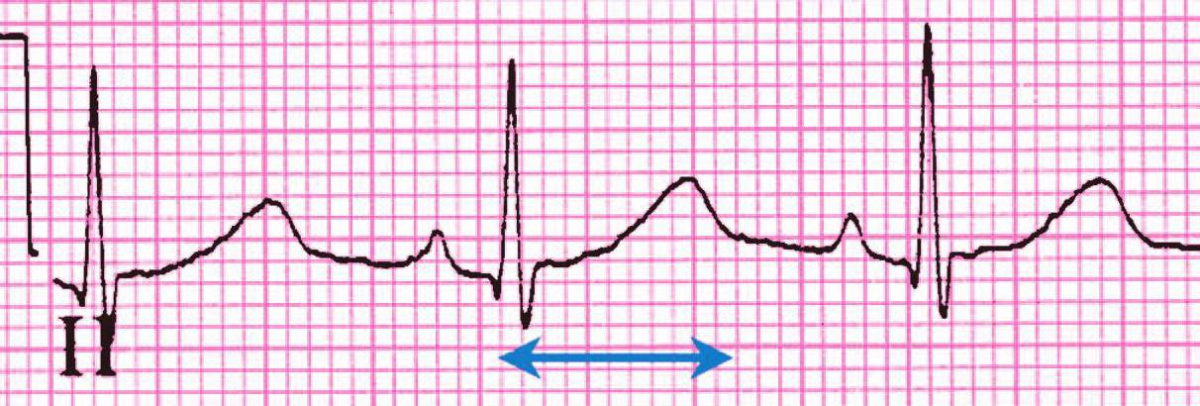
a. QT interval = 0.66, RR interval = 0.85, QTc interval = 0.575
b. QT interval = 0.44, RR interval = 0.76, QTc interval = 0.555
c. QT interval = 0.44, RR interval = 0.76, QTc interval = 0.505
7. Given the QTC interval above 500msec, what arrhythmia is the patient at risk of developing?
a. Torsade de Pointes
b. Pheochromocytoma
c. Type II second degree AV block
8. Which of the following drugs would be unlikely to cause QTc interval prolongation?
a. Quinidine
b. Sotalol
c. Verapamil
9. Which of the following drugs can treat patients with Torsade de Pointes?
a. Magnesium sulfate
b. Amiodarone
c. Adalimumab
10. If a patient is taking sotalol and his renal function markedly decreases, what is likely to happen to the QTc interval?
a. It would get longer
b. It would get shorter
c. It would stay the same