Learning Objectives
After completing this application-based continuing education activity, pharmacists will be able to
- List the pathogenesis of CML
- Discuss the different generations of BCR-ABL TKIs for CML, their places in therapy, and investigational agents in the pipeline for CML
- Explain an appropriate treatment plan for patients with T315I mutation-positive CML including the clinical benefits of molecular monitoring/mutation testing
- Describe the pharmacist’s responsibilities in promoting TKI adherence and management of BCR-ABL TKI adverse effects
After completing this application-based continuing education activity, pharmacy technicians will be able to
- List CML’s primary signs and symptoms
- Recognize BCR-ABL TKIs used to treat CML
- Describe warning signs of adverse reactions and nonadherence in patients taking BCR-ABL TKIs for CML
- Identify OTC products that are likely to interact with TKIs
Release Date:
Release Date: August 15, 2022
Expiration Date: August 15, 2024
Course Fee
FREE
An Educational Grant has been provided by:
Novartis Pharmaceuticals Corporation
ACPE UANs
Pharmacist: 0009-0000-22-050-H01-P
Pharmacy Technician: 0009-0000-22-050-H01-T
Session Codes
Pharmacist: 22YC50-FXP48
Pharmacy Technician: 22YC50-PXF92
Accreditation Hours
2.0 hours of CE
Accreditation Statements
| The University of Connecticut School of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education. Statements of credit for the online activity ACPE UAN 0009-0000-22-050-H01-P/T will be awarded when the post test and evaluation have been completed and passed with a 70% or better. Your CE credits will be uploaded to your CPE monitor profile within 2 weeks of completion of the program. |  |
Disclosure of Discussions of Off-label and Investigational Drug Use
The material presented here does not necessarily reflect the views of The University of Connecticut School of Pharmacy or its co-sponsor affiliates. These materials may discuss uses and dosages for therapeutic products, processes, procedures and inferred diagnoses that have not been approved by the United States Food and Drug Administration. A qualified health care professional should be consulted before using any therapeutic product discussed. All readers and continuing education participants should verify all information and data before treating patients or employing any therapies described in this continuing education activity.
Faculty
Bisni Narayanan, MS, PharmD
Pharmacy Supervisor- Operations
Yale New Haven Health Systems
Hamden, CT
Faculty Disclosure
In accordance with the Accreditation Council for Pharmacy Education (ACPE) Criteria for Quality and Interpretive Guidelines, The University of Connecticut School of Pharmacy requires that faculty disclose any relationship that the faculty may have with commercial entities whose products or services may be mentioned in the activity.
Dr. Narayanan has no relationship with ineligible companies and therefore has nothing to disclose.
ABSTRACT
Chronic myeloid leukemia (CML), a myeloproliferative neoplasm, accounts for approximately 15% of newly diagnosed cases of leukemia in adults. With the advent of tyrosine kinase inhibitors, patients with CML have a life expectancy close to that of the general population. Clinicians face several challenges—selecting initial therapy, monitoring for treatment resistance and failure, determining when to switch therapy—while caring for patients with CML. TKIs are first-line therapy for newly diagnosed Philadelphia chromosome positive chronic phase CML. Second-generation TKIs (bosutinib, dasatinib, and nilotinib) provide a deeper and faster response but have no impact on survival compared to imatinib, a first-generation TKI. The gate keeper mutation T315I in the BCR-ABL1 gene can create resistance to all first- and second-generation TKIs. Ponatinib and asciminib are the only approved TKIs with activity against the T315I mutation. Patient education and drug monitoring are the most profound opportunities for pharmacists.
CONTENT
Content
Introduction
In the mid-1990s Mel Mann, a 37-year-old U.S Army major received some terrible news.1 Just diagnosed with a terminal form of leukemia—chronic myeloid leukemia (CML)—his doctor informed him he had three years to live. Being a Black man, his chances of a bone marrow transplant—the only possible cure at the time— were slim. Very few Black Americans registered in the marrow registry.2 By 1998, Mel Mann had exhausted all available experimental drugs and was growing frail and fatigued by the day. In the summer of 1998, he enrolled in a phase 1 clinical trial. He received STI-571, an experimental drug. Nine months later he ran the 26.2-mile marathon in Alaska, and few months after that cycled 111 miles in Tucson, Arizona. Today, Mel Mann is the longest surviving patient on STI-571— now known as imatinib (Gleevec®).
Imatinib would change oncology’s course, opening an entire new field of precision medicine. In the March 2017 New England Journal of Medicine, an editorial called imatinib “the drug that changed everything.” 3
CML, a myeloproliferative neoplasm, accounts for approximately 15% of newly diagnosed cases of leukemia in adults.4 More than 90% of patients with CML have the Philadelphia (Ph) chromosome and its oncogenic product, BCR-ABL1. In 2022, about 8860 new CML cases will be diagnosed in the United States (U.S.), and roughly 1220 patients will die from the disease.5 The introduction of tyrosine kinase inhibitors (TKIs) that target the BCR-ABL1 oncoprotein has dramatically improved patient survival. Median survival in patients with CML improved from three to six years before the imatinib era to a 10-year overall survival in 83.3% in imatinib-treated patients.4 Patients with CML now achieve lifespans close to normal life expectancies. Complications of CML include hepatomegaly and or splenomegaly, worsening anemia, bleeding or clotting complications from changes in platelets, recurrent infections, bone pain, and fever. Splenomegaly is the most common physical sign in about 20% to 40% of patients.5
BCR-ABL Oncoprotein
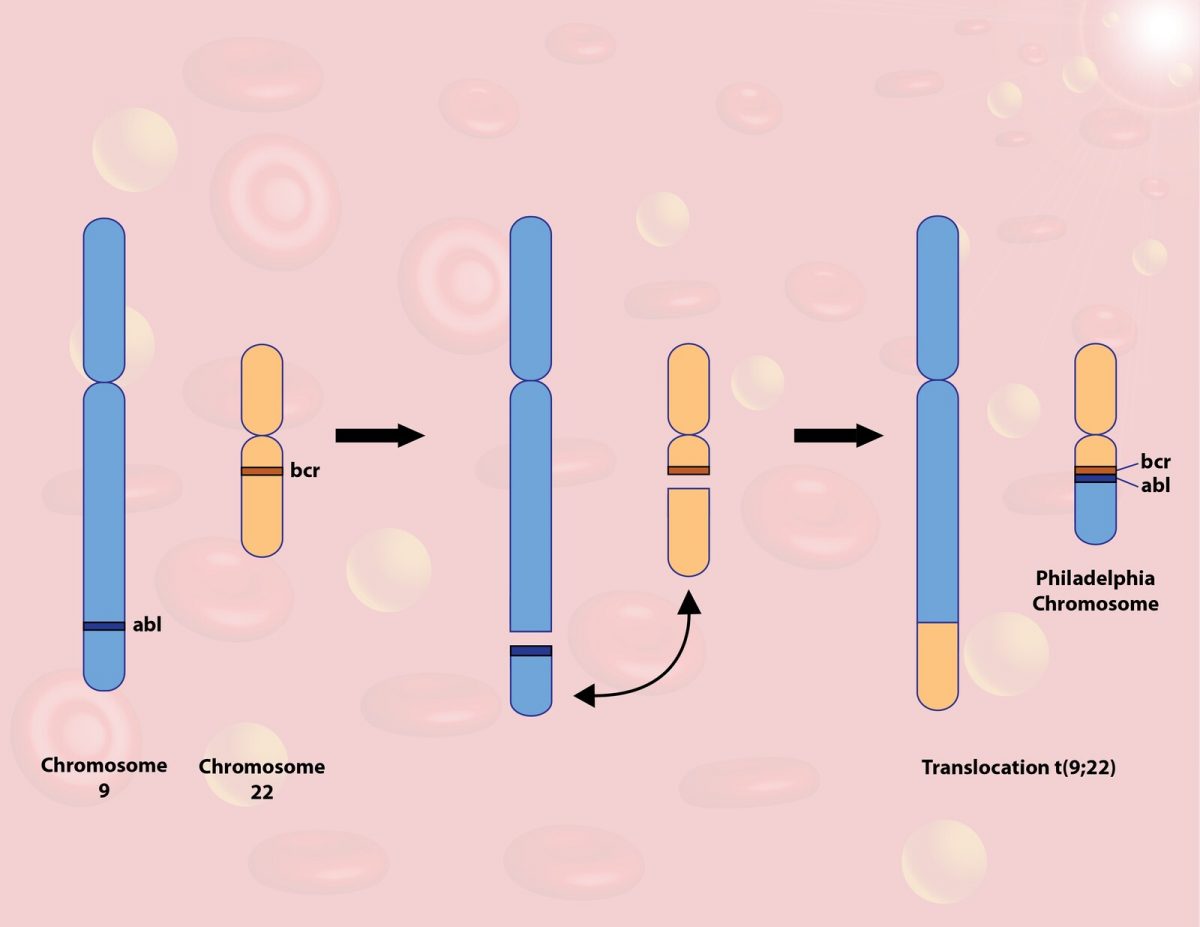
Ph chromosome, named after the city where it was described, is generated during reciprocal translocation between chromosome 9, where the ABL1 gene is located, and chromosome 22, where the breakpoint cluster gene (BCR) is located.6 This results in the formation of the fusion gene BCR-ABL1. BCR-ABL1 generation causes constitutive tyrosine kinase activation leading to continued cell proliferation, cell survival, inhibition of apoptosis (programmed cell death), and activation of transcription factors. Several downstream signaling pathways are activated: JAK2/STAT, PI3K-AKT-mTOR, MAPK/ERK, TRAIL-induced apoptosis, and C/EBP-mediated differentiation. Oncologists employ cytogenetic testing or a reverse transcriptase PCR test to detect the Ph chromosome.
CML Staging
CML is characterized by uncontrolled growth of myeloid cells at different maturation stages and affects peripheral blood and bone marrow. Oncologists classify CML as chronic, accelerated, or blast phase based on the number of blasts (immature blood cells) present in the blood or bone marrow. The initial phase of CML is chronic, with the accelerated or blast phases indicating disease progression. Without medical intervention in the chronic phase, patients progress to acute fatal leukemia within four to six years.7 At diagnosis, 90% to 95% of patients are in the chronic phase. In the U.S., about 50% of patients diagnosed with CML are asymptomatic. Most patients will evolve into the accelerated phase before the blast phase, but about 20% of patients will develop sudden blast crisis without accelerated signs.5
Patients in the chronic phase usually have less than 5% blasts in the bone marrow, are asymptomatic or have mild symptoms, and respond to TKI therapy.8
Patients in the accelerated phase may exhibit one or more of the following5:
- Persistent or increasing high white blood cell count (greater than 10 x 109/L) unresponsive to therapy
- At least 20% basophils in blood
- Blasts 10% to 19% in blood or bone marrow
- Thrombocytopenia/platelet counts lower than 100 x 109/L
- Thrombocytosis/platelet counts higher than 1000 x 109/L, unresponsive to therapy
- Additional chromosomal abnormalities in the Ph+ cells at diagnosis or new clonal chromosomal abnormality in Ph+ cells during therapy
- Persistent or increasing splenomegaly unresponsive to therapy
- Patients may present with worsening anemia, splenomegaly, and organ infiltration
Patients in the blast phase have at least 20% blasts in the peripheral blood and/or bone marrow or extramedullary (outside the bone marrow) blast proliferation.8 Extramedullary disease proliferation occurs when blast cells infiltrate from the bone marrow into the lymph node, skin, spleen, bone or central nervous system and form tumors. Patients may present with worsening constitutional symptoms, bleeding, fever, and infections.5
Response Type in CML
According to the National Cancer Institute, surrogate end points are biomarkers that substitute for clinical end points. In CML, surrogate end points include hematologic, cytogenetic, and molecular response measures that may correlate with a clinical endpoint or outcome.9 In patients with CML, complete hematologic response is typically the first surrogate end point reached. Cytogenetic response measures the decline in disease burden by analyzing the bone marrow Ph+ metaphase chromosome (chromosomes are analyzed in the metaphase as they are easy to visualize then). Oncologists monitor reductions in the number of clonal leukemic cells with real-time quantitative PCR based molecular analysis. Table 1 describes hematologic, cytogenetic, and molecular response monitoring that are key strategies for managing patients with CML.
Table 1. CML Response Type10
| Response Type | Features | Test used to measure response |
| Hematologic
Complete hematologic response (CHR) |
WBC <10 x 109/L, basophils < 5%, platelet count <450 x 109/L, absence of myelocytes, promyelocytes, myeloblasts in differential, spleen nonpalpable | CBC with differential |
| Cytogenetic | ||
| Complete cytogenetic response | No Ph+ chromosome detected | Bone marrow cytogenetics looks at presence of Ph+ chromosome in a bone marrow sample
FISH analysis uses fluorescent dye-labeled probes to light up the BCR-ABL1 sequence |
| Partial cytogenetic response | 1 % to 35 % cells have Ph+ chromosome | |
| Minor cytogenetic response | > 35 % to 65 % cells have Ph+ chromosome | |
| Major cytogenetic response | 0 % to 35 % of cells have Ph+ chromosome | |
| Molecular | ||
| Complete molecular response (CMR) | No BCR-ABL1 gene detected | Quantitative PCR measures the BCR-ABL1 RNA transcripts |
| Major molecular response (MMR) | At least a 3- log reduction in BCR-ABL1 levels or BCR-ABL1 ≤ 0.1% | |
| Early molecular response (EMR) | BCR-ABL1 ≤ 10% at 3 and 6 months | |
| Deep molecular response (DMR) | MR4.0: BCR-ABL1 ≤ 0.01% or MR4.5: BCR-ABL1 ≤0.0032% | |
Surrogate end points may translate to the following clinical outcomes5:
- Early molecular response (BCR-ABL at or below 10% at six months) with complete cytogenetic response 12 months later translates to significantly improved patient survival.
- Major molecular response (BCR-ABL at or below 0.1%) translates to modest improvement in event-free survival, possible longer duration of complete cytogenetic response, however major molecular response does not translate to survival benefit.
- Deep molecular response translates to a possibility of therapy discontinuation (discussed in detail below).
PAUSE AND PONDER: Can you list all of the currently available TKI inhibitors and sort them by generation? Why does the generation matter?
TYROSINE KINASE INHIBITORS IN CML
TKIs are targeted therapies that inhibit the action of tyrosine kinase— an enzyme implicated in cell signaling, proliferation, differentiation, and programmed cell death. In CML, TKIs target the BCR-ABL1 oncoprotein which causes uncontrolled cell growth.
Clinical considerations for first-line therapy in chronic phase CML
The key goal in chronic phase CML is to achieve clinical remission, maintain long-term disease control, and avoid disease progression to accelerated and blast phases while limiting treatment related adverse effects.11 For patients who achieve sustained deep molecular remission, discontinuation of therapy with treatment-free remission is a long-term goal.
Currently, four TKIs are Food and Drug Administration (FDA)-approved for chronic phase CML frontline therapy: bosutinib, dasatinib, imatinib, and nilotinib (See Figure 1). Prescribers select a BCR-ABL1 TKI for initial treatment of chronic phase CML based on the CML risk score.11 Providers use a validated CML scoring system—Sokal, Euro, or EUTOS (European Treatment and Outcome Study) long-term survival score (ELTS) scoring systems—to stratify patients into low, intermediate, and high risk.
- The Sokal risk calculation uses age, spleen size, platelet count, and percentage of myeloblasts in the peripheral blood.
- The Euro risk calculation uses age, spleen size, platelet count, and percentage of blasts, eosinophils, and basophils in the peripheral blood. See how to do the calculation here: https://www.leukemia-net.org/content/leukemias/cml/euro__and_sokal_score/index_eng.html.
- The ELTS score employs age, spleen size below costal margin, blasts in peripheral blood, and platelet count. Clinicians evaluate other variables as well at diagnosis prior to any CML therapy. The online calculator can be found here: ELTS-Score (leukemia-net.org).
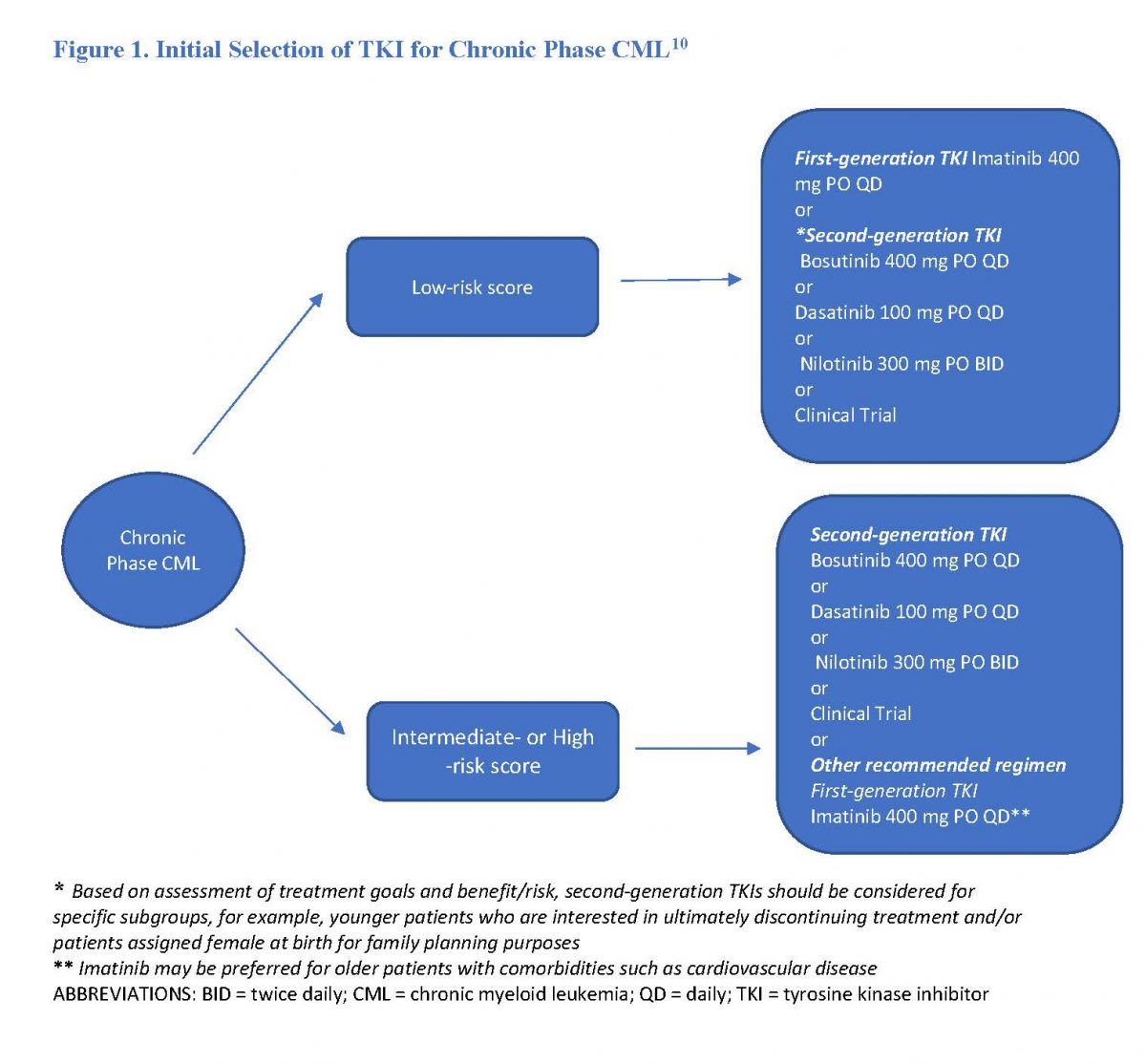
Disease progression is more likely in patients in the intermediate- and high-risk strata. Providers must consider cost, availability, adverse effect profile, drug interactions, comorbid illness, and patient preference in choosing an initial TKI.11 Researchers have reported a faster molecular response and higher rate of major molecular response (BCR-ABL1 at or below 0.1%), and deep molecular response (BCR-ABL1 at or below 0.0032%) among patients treated with a second-generation TKI across all risk scores.5 In some patients, this may facilitate subsequent therapy discontinuation. Hence, second-generation TKIs may be an option for younger patients, especially women for fertility purposes.5
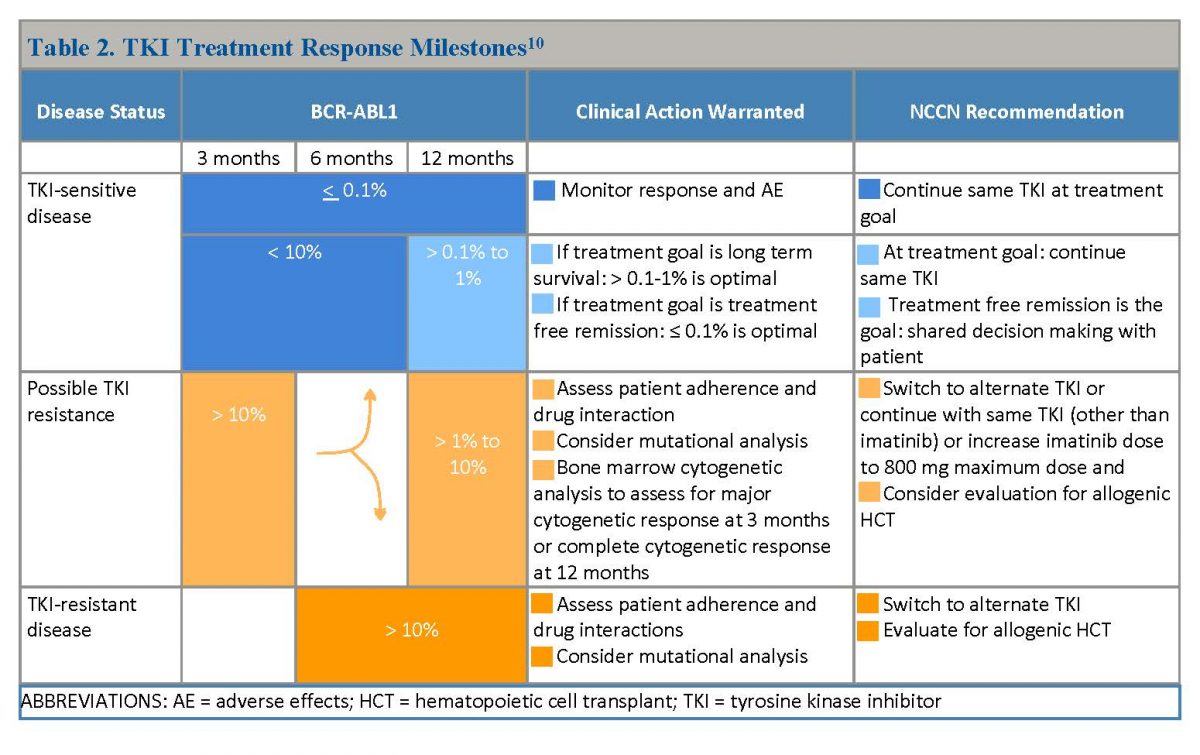
Treatment Response
At baseline, all patients with CML undergo a bone marrow examination to assess the percentage of blasts and basophils.5 Experts prefer the quantitative PCR (qPCR) test to monitor response to TKI therapy, and monitor patients every three months, including those meeting response milestones at three, six, and 12 months (See Table 2). Patients achieving complete cytogenetic response are monitored every three months for two years, and every three to six months thereafter. All FDA-approved TKIs are oral, but despite the convenience of oral therapy, about one-third of patients are nonadherent. Adherence is associated with better clinical outcomes and prognosis. Patients achieving complete cytogenetic response within 12 months after first-line TKI therapy have better long-term survival.5
Table 2. TKI Treatment Response Milestones10
FRONTLINE TREATMENT OPTIONS
The frontline treatment options for patients with chronic phase CML are first- or second-generation TKIs. Oncologists choose between them based on individual patient characteristics such as age, disease state, and comorbidities.
First-Generation TKI
The FDA approved imatinib, the only first-generation TKI, for the treatment of newly diagnosed adult patients with Ph+ CML in the chronic phase and for Ph+ CML in blast crisis, accelerated phase, or in chronic phase after failure of interferon-alpha therapy.12 In the pediatric population, imatinib is approved for newly diagnosed Ph+ CML in chronic phase or disease resistant to interferon therapy. Imatinib inhibits the BCR-ABL1 tyrosine kinase by competitively inhibiting the ATP-binding site, resulting in protein phosphorylation inhibition in the downstream signal transduction pathway inducing apoptosis in BCR-ABL1 positive cell lines.
The recommended dose for adult patients is 400 mg/day in the chronic phase, and 600 mg/day in the accelerated or blast crisis.12 Patients in the accelerated or blast crisis may receive 800 mg/day (400 mg twice daily) if they have no severe adverse reactions. The recommended dose for children with newly diagnosed Ph+ CML is 340 mg/m2/day (not to exceed 600 mg).12
Imatinib can cause edema/weight gain, fatigue, peripheral and periorbital edema, bone and muscle aches, and nausea.5 With long term therapy, up to 10% of patients experience creatinine elevations. Rare neurologic complications of worsening of Parkinsonism, and dementia-like findings have also been reported.
In 2002, a group of French physicians reported an intriguing side effect among patients treated with imatinib—changes in hair repigmentation. Patients who had grey hair before treatment had progressive repigmentation (head and body) after imatinib treatment initiation.13 Investigators have also reported blue grey skin discoloration among patients treated with imatinib.14 One proposed mechanism is along with inhibiting tyrosine kinase activity, imatinib also inhibits c-KIT, a protein expressed in melanocytes, and skin basal cells.
Patients treated with imatinib are slower to achieve cytogenetic and molecular response than those treated with second-generation TKIs, but long-term outcomes are comparable.5 High-dose imatinib (800 mg) and imatinib-based combinations (with interferon) should not be used as frontline therapy for Ph+ chronic phase CML. Patients in a high-dose study group achieved faster complete cytogenetic and major molecular response compared to the standard dose group, but the rates were not significantly different at 12 months.5
Dose Reduction for Hematologic Adverse Effects
For patients at a starting dose of 400 mg/day for chronic phase CML, if the ANC is less than 1.0 x 109/L and/or platelets are less than 50 x 109/L12
- Stop imatinib until ANC reaches at least 1.5 x 109/L and platelets reach at least 75 x 109/L
- Resume treatment at original starting dose of 400 mg
- If recurrence occurs, stop imatinib and resume at reduced dose of 300 mg
For patients in the accelerated and blast phase at a starting dose of 600 mg/day, if ANC is less than 0.5 x 109/L and/or platelets are less than 10 x 109/L12
- Check if cytopenia is disease related
- If not, reduce imatinib dose to 400 mg
- If cytopenia persists for two weeks, reduce dose to 300 mg
- If cytopenia persists for four weeks, and is unrelated to leukemia stop imatinib until ANC reaches at least 1 x 109 /L and platelets reach at least 20 x 109 /L and then resume treatment at 300 mg
Second-Generation TKIs
Patients may develop imatinib resistance by developing mutations in the BCR-ABL1 gene. Second-generation BCR-ABL TKIs (nilotinib, dasatinib, and bosutinib) have inhibitory activity against the majority of imatinib-resistant mutations.
PAUSE and PONDER: What are the most common reasons for patients to need dose reductions?
Nilotinib
Nilotinib is approved for treatment of newly diagnosed chronic phase Ph+ CML in adult patients resistant to or intolerant to prior therapy that included imatinib.15 The initial dose is 300 mg twice daily approximately 12 hours apart without food. Patients should not consume food for at least two hours before and 1 hour after each dose. Nilotinib’s labeling carries a boxed warning for QT prolongation and sudden death. Patients initiating nilotinib should have an ECG at baseline, seven days after initiation, periodically thereafter, and following dose adjustments. Nilotinib is contraindicated in patients with hypokalemia, hypomagnesemia, or long QT syndrome. Prescribers should correct the patient’s hypokalemia and hypomagnesemia before treatment starts and measure electrolytes periodically during treatment.15
Prescribers should avoid nilotinib or use it cautiously in patients with diabetes or history of pancreatitis since it is associated with hyperglycemia.5 Due to the risk of arterial occlusive events, patients with coronary, cerebrovascular, or peripheral artery disease should limit or avoid nilotinib. Imatinib and bosutinib (discussed below) are safer options in patients with a significant past history of arterial occlusive events.5
In its phase 3 trial, nilotinib-treated patients responded earlier, better and were less likely to progress to accelerated or blast phases than imatinib-treated patients.16 At five-year follow-up, 54% of patients on nilotinib 300 mg dosing twice daily achieved a molecular response of 4.5 (BCR-ABL ≤0.0032%) compared to 31% of patients in the imatinib 400 mg once daily dosing arm.
Dose reduction for hematologic adverse effects.15 For patients at a starting dose of 300 mg twice daily for chronic or accelerated phase CML
- If the ANC is less than 1.0 x 109 /L and/or platelet count is less than 50 x 109 /L, stop nilotinib and monitor blood counts
- If ANC exceeds 1.0 x 109 /L and platelet count exceeds 50 x 109 /L, resume at prior dose within two weeks
- If blood counts do not recover for at least two weeks, reduce dose to 400 mg once daily
Dose adjustments for QT prolongation. If patient’s ECG shows QTc exceeds 480 msec
- Stop nilotinib, check serum potassium and magnesium, and if below lower limit of normal correct to within normal limits. Review concomitant medications.
- Resume within two weeks if QTc return is less than 450 msec, and to within 20 msec of baseline
- If QTc is between 450 and 480 msec after two weeks, reduce dose to 400 mg once daily
- Discontinue nilotinib if following dose reduction to 400 mg once daily, QTc returns to exceeding 480 msec
Dose reduction for non-hematologic abnormalities. For elevated serum lipase or amylase at Grade 3 or worse, or bilirubin or hepatic transaminases at or above Grade 3
- Withhold nilotinib, and monitor serum lipase or amylase, bilirubin or hepatic transaminases
- Resume treatment at 400 mg once daily once lab abnormalities return to Grade 1 or lower
Dasatinib
Dasatinib is approved for newly diagnosed adults with Ph+ CML in chronic phase, and for adults with chronic, accelerated, myeloid, or lymphoid blast phase Ph+ CML with resistance or intolerance to prior therapy including imatinib.17
The recommended dose of dasatinib for chronic phase CML is 100 mg once daily and for accelerated or blast phase CML, the dose is 140 mg once daily.17 In clinical trials, treatment with dasatinib was associated with severe myelosuppression and severe bleeding-related events from thrombocytopenia. Investigators reported CNS and gastrointestinal (GI) symptoms as well as other cases of severe hemorrhage in patients. Prescribers should use dasatinib with caution in patients on concomitant platelet inhibitors or anticoagulants. In clinical trials, pleural effusions were also more common in the dasatinib groups. In patients with histories of lung disease (e.g., chronic obstructive pulmonary disease), cardiac disease (e.g., congestive heart failure), or uncontrolled hypertension, prescribers should avoid dasatinib. Prescribers should consider an alternative TKI for patients with preexisting pulmonary arterial hypertension, since it is a rare complication with dasatinib treatment.17
In early clinical trials, many patients experienced adverse effects at the 100 mg daily dose, resulting in discontinuation of the drug.5 Subsequent studies with dasatinib 50 mg daily and dasatinib taken five days/week have shown good remission rates with minimal toxicity. Researchers recently reported that dasatinib at 20 mg daily is safe for patients older than 70 years old with medical comorbidities. Patients achieved molecular remissions with a lower incidence of adverse events compared to the higher dose (100 mg daily dose).18
Dose reduction for hematologic adverse effects.15 For patients taking 100 mg once daily for chronic phase, if the ANC is less than 0.5 x 109 /L and/or platelet count is less than 50 x 109 /L
- Stop dasatinib until ANC is at least 1 x 109 /L and/or platelet counts are at least 50 x 109/L
- Resume treatment at original dose if blood counts recover within seven days
- If platelets are less than 25 x 109 /L or ANC is less than 0.5 x 109 /L for more than seven days, follow step 1. Resume dasatinib at a reduced dose of 80 mg once daily for the second episode. If a third episode occurs, for newly diagnosed patients, further reduce dose to 50 mg once daily; for patients resistant or intolerant to prior therapy including imatinib, discontinue dasatinib.
Bosutinib
Bosutinib is approved for newly diagnosed chronic phase Ph+ CML and chronic, accelerated or blast phase CML with resistance or intolerance to prior therapy.19 The recommended dose for newly diagnosed CML patients is 400 mg orally once daily with food. For patients with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy, the recommended dose is 500 mg orally once daily with food. If patients do not reach a complete hematologic, cytogenetic, or molecular response and have no adverse effects graded at grade 3 or higher, providers can increase dose to a maximum of 600 mg once daily. Prescribers should monitor patients for GI toxicity (nausea, vomiting, diarrhea, and abdominal pain). Patients with inflammatory bowel disease or renal dysfunction should avoid bosutinib.19 Pharmacists should counsel patients to start bosutinib at 100 mg daily for 1 week, followed by 100 mg dose escalation every seven days until a dose of 400 mg daily is reached to avoid diarrhea.5
Dose reduction for non-hematologic adverse effects.19 For elevated liver transaminases greater than five times the upper limit of normal (ULN), hold bosutinib until recovery to at or less than 2.5 times the ULN and resume at 400 mg once daily thereafter. If recovery takes longer than four weeks, discontinue bosutinib.
For grade 3-4 diarrhea, withhold bosutinib until recovery to grade 1 or lower. Resume bosutinib at 400 mg once daily
Dose reduction for hematologic adverse effects. For patients at starting dose of 400 mg once daily for chronic phase, if the ANC is less than 1 x 109 /L and/or platelet count is less than 50 x 109 /L
- Withhold bosutinib until ANC is at least 1 x 109 /L and/or platelet count is at least 50 x 109 /L
- Resume treatment at same dose if recovery occurs within two weeks.
- If blood counts remain low for more than two weeks, upon recovery reduce bosutinib dose by 100 mg
- Upon recurring cytopenia, reduce dose by an additional 100 mg upon recovery and resume treatment
TKI RESISTANCE
Since imatinib’s introduction, CML’s therapeutic landscape has changed vastly. Patients with CML are developing more drug resistance due to widespread use of TKIs. About 25% of patients with CML will switch TKIs at least once in their lifetime owing to drug resistance or intolerance.20 TKI resistance in patients may be primary or secondary. Primary resistance is the failure to achieve time-dependent endpoints of complete hematologic response, complete cytogenetic response, and major molecular response on TKI treatment initiation. Secondary or acquired resistance is the loss of response following an initial response. Oncologists classify TKI resistance in CML as BCR-ABL1 independent or dependent mechanisms. Patients with primary resistance are more likely to have a BCR-ABL1 independent mechanism, while patients with secondary resistance usually have a BCR-ABL1 dependent mechanism.20
In patients exhibiting secondary resistance, the most effective approach is to use an alternate TKI.20 Healthcare providers must always assess for adherence and drug interactions before switching therapy. Point mutations in the kinase domain of the BCR-ABL1 are the most common mechanism of secondary resistance. Second-generation TKIs overcome most of the mutations that confer resistance to imatinib. However, a gatekeeper mutation T315I (isoleucine replaces threonine in position 315 of BCR-ABL1) is resistant to all currently available TKIs except ponatinib and asciminib. Between 4% and 20% of patients who are resistant to TKI exhibit this gatekeeper mutation.20
In a systematic review of patients with CML with resistance or intolerance to second-generation TKIs, treatment with a different second-generation TKI in the third-line setting was associated with a lower response rate when compared to the third-generation TKI ponatinib.21
PAUSE AND PONDER: What are the advantages of the third-generation TKIs, and how would you explain them to a patient?
THIRD-GENERATION TKIs
Some patients will become resistant to a second-generation TKI due to development of T315I gatekeeper mutation. Currently, two third-generation TKIs—ponatinib and asciminib— are approved for patients exhibiting this mutation.
Ponatinib
Ponatinib is 500 times more potent than imatinib at inhibiting the BCR-ABL1.22 Ponatinib is not indicated for patients with newly diagnosed chronic phase CML. The FDA approved it for the treatment of three conditions:
- Chronic phase CML with resistance or intolerance to at least two prior TKIs
- Accelerated or blast phase CML for which no other kinase inhibitors are indicated
- T315I-positive CML in chronic, accelerated, or blast phase
The recommended starting dose is 45 mg orally once daily with reduction to 15 mg upon achievement of BCR-ABL1 lower than 1%. If patients do not achieve hematologic response by three months, providers must discontinue the medication.22
The FDA approved ponatinib based on the phase 2 PACE trial (N=449), which evaluated ponatinib’s efficacy and safety in Ph+ acute lymphoblastic leukemia and CML patients (N = 270).23 Patients were eligible for the trial if they had resistance or intolerance to dasatinib or nilotinib or had CML with T315I mutation. Trial participants received ponatinib 45 mg once daily and were stratified by mutation status and disease phase. Among the 267 patients with CML in the chronic phase, 56% achieved major cytogenetic response by 12 months, which included 70% (45/64) of patients with a T315I mutation. At 5-year follow up, 82% of responders maintained the major cytogenetic response with the estimated overall survival rate of 73%.23
In 2013, the manufacturer temporarily withdrew ponatinib from the market following high frequency of severe vascular events.24 One year later, the manufacturer reintroduced the drug with specific dose-reduction recommendations. Ponatinib carries a boxed warning for arterial occlusive events, venous thromboembolic events, heart failure, and hepatotoxicity. Pharmacists should counsel patients on the signs and symptoms of blood clots, heart failure, cardiac arrhythmia, hepatotoxicity, hypertension, pancreatitis, and ocular toxicity.22
Providers must discontinue ponatinib in patients with the following adverse effects22:
- Grade 3 or 4 cardiovascular or cerebrovascular events
- Grade 4 venous thromboembolism
- Grade 4 heart failure
- AST or ALT at least three times the ULN with bilirubin exceeding two times the ULN and alkaline phosphatase less than two times the ULN
- Symptomatic pancreatitis, and serum lipase exceeding five times the ULN
Dose reduction for myelosuppression.22 For patients with ANC less than 1 x 109/L or platelets less than 50 x 109/L, withhold ponatinib until ANC resolves to 1.5 x 1 x 109/L and platelets at least 75 x 109/L, then resume at the same dose.
Upon recurrence, withhold ponatinib until resolution and resume at next lower dose.
Recommended dose reduction for adverse reactions.22 Should adverse events occur, ponatinib’s labeling recommends a first dose reduction to 30 mg orally once daily; a second dose reduction to 15 mg orally once daily; a third dose reduction to 10 mg orally once daily; and discontinuing the drug if patients are unable to tolerate 10 mg once daily.
Asciminib
In 2021, the FDA approved asciminib for the treatment of patients with Ph+ CML in chronic phase previously treated with two or more TKIs, and Ph+ CML with T315I mutation.25 Asciminib has a unique mechanism of action—it binds to the myristoyl site rather than the ATP binding site of the kinase domain like other TKIs.26 Binding to the myristoyl site locks the BCR-ABL1 protein in the inactive conformation and prevents downstream signal transduction events. Asciminib targets both wild type (the version that predominates in natural or normal populations) and mutant BCR-ABL1 including the T315I mutant. The labeling recommends 80 mg orally once daily or 40 mg twice daily for Ph+ CML in chronic phases, and for Ph+ CML with T315I mutation, 200 mg orally twice daily. Pharmacists should counsel patients to avoid food for at least two hours before and one hour after taking asciminib.26
The multi-center open label CABL001X2101 study evaluated asciminib’s efficacy in patients with Ph+ CML with T315I mutation.27 A total of 52 patients (31 patients had prior ponatinib exposure, of whom 15 had ponatinib resistance) with T315I mutation received asciminib at 200 mg orally twice daily. At week 24, 57.1% of ponatinib naïve patients and 28.6% of ponatinib pretreated patients achieved major molecular response. The estimated cumulative major molecular response rate at week 60 increased to 66% and 32% respectively in ponatinib naïve and pretreated patients respectively.
Recommended dose reduction for asciminib.26 For patients with chronic phase CML previously treated with at least two TKIs, the first dose reduction is to 40 mg once daily or 20 mg twice daily. If patients are unable to tolerate 40 mg once daily or 20 mg twice daily, discontinue medication.
For patients with chronic phase CML with T315I mutation, the first dose reduction is to 160 mg twice daily. If patients are unable to tolerate 160 mg twice daily, discontinue medication.
Dose reduction for myelosuppression. For patients with ANC less than 1.0 x 109 /L and/or platelets less than 50 x 109 /L, withhold asciminib until ANC is at least 1 x 109 /L and/or platelets are at least 50 x 109 /L. If blood counts recover within two weeks, resume asciminib at the starting dose. If blood counts take more than two weeks to recover, reduce the asciminib dose.
Currently, no randomized head-to-head trials compare ponatinib with asciminib in patients with CML.5
CML AGENTS IN THE PIPELINE
Table 3 summarizes the many entities that are in the pipeline and have potential to treat CML.
Table 3. The CML Development Pipeline28
| Agent and Clinical Trial Phase | TKI therapy | Mechanism of action | Indication |
| ELVN-001 in Phase 1
|
No | Phosphotransferase inhibitor; oral | CML with or without T315I mutations in patients who are relapsed, refractory or intolerant to TKIs |
| Flumatinib in Phase 3 | Yes | BCR-ABL1 TKI; oral | Newly diagnosed CML |
| IkT-001Pro in preclinical | Yes | Prodrug formulation of imatinib; route currently N/A | Stable phase CML |
| KDS-1001 in Phase 1 | No | Natural killer cell replacement; IV | CML with persistent or recurrent molecular residual disease after one year of TKI therapy |
| KRT-232 in Phase 1b/2 | No | MDM2 (murine double minute 2) inhibitor, leading to p53 mediated transcription inhibition; oral | Relapsed or refractory Ph+ CML in chronic phase |
| Lonafarnib in Phase 1 | No | Aurora kinase inhibitors; oral | Chronic phase CML or accelerated CML after failure of imatinib |
| Motixafortide in Phase 2 | No | CXCR4 receptor antagonist; Subcutaneous | |
| Olverembatinib, approved in China | Yes | Third-generation BCR-ABL1 TKI; oral | TKI-resistant CML or accelerated phase CML with T315I mutation |
| Radotinib in Phase 3/2 | Yes | BCR-ABL1 TKI; oral | Newly diagnosed CML, or accelerated CML with resistance or intolerance to imatinib |
| Tipifarnib in Phase 1 | No | Aurora kinase inhibitor; oral | Chronic phase CML after failure of imatinib |
| Vodobatinib in Phase 1/2 | Yes | BCR-ABL1 TKI; oral | CML patients ≥ 3 agents |
TREATMENT FREE REMISSION
In CML, treatment free remission refers to patients having a stable deep molecular response without ongoing TKI treatment.10 With CML patients approaching a life expectancy to that of the general population, the focus has shifted to quality of life and cost savings. According to the National Comprehensive Cancer Network (NCCN) guidelines, certain patients may discontinue TKI10:
- Patients in chronic phase CML with no prior history of accelerated or blast phase
- Patients who have taken approved TKI therapy for at least three years, with deep molecular response (BCR-ABL1 at or below 0.01%) for at least two years as documented on at least four tests performed at least three months apart
Upon TKI discontinuation, healthcare providers must continue to monitor patients closely with monthly molecular monitoring for the first six months, followed by bimonthly tests during months seven through 12, then quarterly for patients who remain in major molecular response.10 If patients exhibit a loss of major molecular response, they must resume TKI within four weeks with monthly molecular monitoring until a major molecular response is re-established. These patients must undergo molecular monitoring every three months indefinitely. If patients do not achieve major molecular response within three months of TKI resumption, healthcare providers must check for kinase domain mutations.10
Clinical studies have only studied the feasibility of treatment free remission following discontinuation of dasatinib, imatinib, and nilotinib.10 However, the NCCN guidelines do indicate it would be reasonable to extrapolate the results irrespective of the TKI in patients who have achieved and maintained deep molecular response for at least two years.10
PAUSE AND PONDER: Which OTC drugs might be a problem for patients who take TKIs? How might you remember to screen for those drugs? What alternatives would patients be able to use?
THE PHARMACY TEAM’S RESPONSIBILITIES
All TKIs are metabolized by the cytochrome P450 system. These drugs have significant potential for drug-drug interactions with some drug interactions causing toxicity or affecting therapeutic response.
Asciminib26
With this TKI, pharmacists should screen for strong CYP3A4 inhibitors, CYP3A4 substrates, CYP2C9 substrates, and P-glycoprotein (P-gp) substrates.
- Monitor closely for adverse effects with strong CYP3A4 inhibitors with asciminib 200 mg twice daily.
- Avoid use of asciminib 200 mg twice daily with CYP3A4 substrates.
- Avoid concomitant asciminib at all doses with CYP2C9 substrates. If asciminib use is unavoidable, reduce the CYP2C9 substrate dose and limit the asciminib to 80 mg daily.
- Consider alternative therapy with non-CYP2C9 substrate with asciminib 200 mg twice daily.
- Closely monitor for adverse reactions during concomitant P-gp substrates and asciminib at all recommended doses.
Bosutinib19
Avoid concurrent use of bosutinib with strong or moderate CYP3A inhibitors and inducers
Dasatinib17
Dasatinib is a CYP3A4 substrate and may interact with CYP3A4 inhibitors or inducers.
Imatinib12
CYP3A4 inducers may decrease imatinib levels, and conversely, CYP3A4 inhibitors may increase imatinib levels. Imatinib inhibits CYP3A4 and may increase the Cmax and AUC of other drugs. Patients who require anticoagulation should receive low-molecular weight or standard heparin (not warfarin). Co-administration with acetaminophen may increase the systemic exposure of acetaminophen.
Nilotinib15
CYP3A4 inhibitors and inducers may affect nilotinib’s serum concentration. Nilotinib inhibits CYP3A4, CYP2C8, CYP2C9, and CYP2D6. It may also induce CYP2B6, CYP2C8 and CYP2C9. Therefore, nilotinib may alter other drugs’ serum concentrations.
Ponatinib22
Avoid concurrent use or reduce the ponatinib dose if strong CYP3A inhibitors cannot be avoided; avoid concurrent use with strong CYP3A inducers.
Table 4 lists common medications that may alter the TKI’s therapeutic effect. Many of these are over the counter, so pharmacy technicians can alert the pharmacist if patients who take TKIs purchase these products so they can explore appropriate alternatives.
Table 4. TKI Drug Interactions with Common Drugs and Supplements10
| Drug Class | Effect |
| Proton Pump Inhibitors (PPIs) | Decreased exposure of bosutinib, dasatinib, nilotinib, ponatinib
No major interaction with imatinib |
| H2 receptor antagonists (H2RAs) | Avoid H2RAs with bosutinib, dasatinib, nilotinib.
If absolutely necessary consider once-daily H2RAs ≥2 hours after bosutinib or dasatinib, or ≥2 hours after or ≥10 hours before taking nilotinib No major interaction with imatinib and ponatinib |
| Antacids | Decreased exposure of bosutinib, dasatinib, nilotinib, and ponatinib.
For above TKIs, may use antacids at least 2 hours before or at least 2 hours after taking TKIs No major interaction with imatinib |
| Antidepressants
· Fluoxetine · Bupropion · Citalopram |
Minor increase in exposure of bosutinib, dasatinib, imatinib, and ponatinib. Monitor QTc for these TKIs.
Avoid with nilotinib due to QTc prolongation risk |
| Cardiovascular medications
· Amiodarone · Diltiazem · Verapamil
|
Bosutinib, dasatinib, imatinib, ponatinib: Increase in exposure and arrhythmia risk. Strongly consider alternative cardiac medication or TKI dose adjustment
Avoid nilotinib |
| Anti-infectives
· Oral azole antifungals o Fluconazole o Voriconazole · Clarithromycin · Telithromycin · Ritonavir |
Increase in exposure of all TKIs. Strongly consider alternative anti-infective or TKI dose adjustment |
| Fluoroquinolones
· Levofloxacin · Ciprofloxacin |
QTc monitoring for bosutinib, dasatinib
Use nilotinib with caution No major interactions with imatinib and ponatinib |
| Herbal supplements
· Curcumin |
Increase in exposure of all TKIs, discontinue supplement |
| Herbal supplements
· St John’s Wort |
Avoid use with all TKIs, decrease in TKI exposure |
Pharmacists must also be aware of the common class adverse effects and offer patients appropriate management strategies as follows:8,29
Cardiovascular considerations. Patients with pre-existing peripheral vascular disease or cardiovascular disease should avoid nilotinib. Patients with hypokalemia or hypomagnesemia or at risk of QT prolongation syndrome should avoid dasatinib and nilotinib. Bosutinib or imatinib can be considered for patients with a high number of risk factors for cardiovascular disease of history of arrythmias or heart disease.
All TKIs for CML are associated with increased bleeding, but the risk is more pronounced with dasatinib than other TKIs. Ponatinib has an associated incidence of heart failure and QT interval shortening. Pharmacists should check to ensure patients taking ponatinib are being monitored for cardiac function. They should also check to ensure potassium and magnesium levels are acceptable before initiating the BCR-ABL1 TKIs.
Patients experiencing fluid retention may take diuretics to reduce edema and the clinical team should monitor their symptoms closely.
Pulmonary considerations. Dasatinib carries a risk for pleural effusions and pulmonary hypertension. In patients with a history of lung disease or with risk of developing pleural effusions, the guidelines prefer bosutinib, imatinib, or nilotinib. If patients develop a cough, shortness of breath, or chest pain during dasatinib therapy, the team should order a chest X-ray. Pharmacy technicians are ideally positioned to recognize these symptoms and alert the pharmacist for patients on a BCR-ABL1 TKI.
Patients may take diuretics or short course of steroids for management of pleural effusions. Providers may also dose reduce and/or interrupt treatment to manage pleural effusions.
Metabolic considerations. Nilotinib increases serum lipids and glucose. If patients need a statin medication, prescribers should select one not metabolized by the CYP3A4 (pravastatin and rosuvastatin) to avoid drug interactions with nilotinib. In patients with a history of diabetes or history of pancreatitis, bosutinib, dasatinib, or imatinib may be used.
GI considerations. Prescribers should use dasatinib with caution in patients with ulcerative colitis, or GI ulcers due to risk of hemorrhage. Refer to Table 4 for potential interactions with commonly used PPIs, antacids, and H2RAs.
Advising patients to take the medication with or without food and/or water in accordance with each drug’s labeling helps avoid GI effects. Patients may take antinausea, antidiarrheal, or antiemetic medications for symptom relief.
Hepatic and renal considerations. Consider dasatinib for patients with liver disease, as hepatotoxicity is least pronounced with dasatinib. Ponatinib has the highest risk for developing high-grade transaminase elevations. For patients with renal impairment, asciminib, dasatinib and nilotinib require no dose adjustment, but imatinib and bosutinib may require dose reductions. Drugs that alter liver metabolism by cytochrome P450 enzymes and acetaminophen should be used with caution or avoided entirely—prompt treatment with glucocorticoids in severe cases of hepatotoxicity assists in hepatic recovery.
For elevated liver enzymes, providers must check liver function regularly and may consider dose reduction and/or interruption.
Myelosuppression. Myelosuppression is common with all TKIs usually in the first few weeks to months of therapy. Providers must routinely monitor blood chemistry and may consider administration of growth factors as required. Pharmacy technicians with access to the electronic health record can remind patients about their routine blood test schedule.
Dermatologic effects. Rash is common with all TKIs. Most cases are dose-related and self-limiting. Patients may use topical or systemic steroids or antihistamines or reduce dose and/or interrupt treatment to manage dermatologic effects. Pharmacy technicians are often the first point of patient contact at the pharmacy. They should refer patients who are on BCR-ABL1 TKIs seeking advice on OTC medications to the pharmacist.
Pregnancy considerations. Female patients must stop TKI therapy prior to conception and during pregnancy. Patients must be in sustained MMR for at least two years before attempting to conceive. Interferon alfa-2a or peginterferon alfa-2a is an option for patients presenting with CML during pregnancy. Patients may restart TKI therapy after delivery. Pharmacists must counsel patients not to breastfeed as TKIs pass into breast milk.
CONCLUSION
Patients with CML have lifespans approaching the general population with treatment. They have multiple options of first-, second-, and third-generation TKIs— imatinib, dasatinib, nilotinib, ponatinib, bosutinib, and asciminib. Providers must choose the appropriate treatment based on patient and disease characteristics. Pharmacists must be aware of the BCR-ABL1 TKIs side effect profile as adverse effects negatively impact the patient’s quality of life leading to non-adherence. Patients with T315I mutation, who have failed first- and second-generation TKIs now have two options — ponatinib, and asciminib. Asciminib has a unique mechanism of targeting the ABL myristoyl pocket. Although the treatment landscape of CML has changed vastly in the last few years, nonadherence is still an issue among patients. Pharmacists and pharmacy technicians must emphasize the importance of 100% adherence for optimal patient outcomes.
UConn OPPD thanks Brian Pelletier, PharmD, for his assistance with graphics.
Pharmacist Post Test (for viewing only)
Pharmacists Post-test
Patient AA is a 60-year-old man with a past medical history of hypertension and type 2 diabetes. His medication history includes metformin, and valsartan/hydrochlorothiazide. He presents with months of fatigue and 10-pound weight loss. He is diagnosed with CML and initiated on a BCR-ABL TKI. His blood pressure is controlled by medication.
Blood work: WBCs 39180/mcL, Platelets 216000/mcL, 1% blasts, 4% basophils, 3% eosinophils
1. Identify the phase of CML in which AA would be classified?
a) Chronic Phase
b) Accelerated Phase
c) Blast Phase
2. The oncology APRN calls to alert you that she is sending a prescription for nilotitinb. What should you tell her?
a) The lead time to order and receive nilotinib is several weeks.
b) Nilotinib is not the best choice in patients with diabetes.
c) Be certain to check this patient’s serum lipids and glucose annually.
3. AA was initiated on a BCR-ABL TKI and approximately 24 months after initiation, presented to the pharmacy with shortness of breath and cough. You refer the patient to his oncologist. Chest x-ray reveals pleural effusion. Which BCR-ABL TKI is likely to cause this adverse effect?
a) Dasatinib
b) Nilotinib
c) Imatinib
4. Three months after initiating therapy, quantitative PCR test shows BCR-ABL transcripts at 3.2%. What type of response is AA exhibiting?
a) Complete molecular response
b) Major molecular response
c) Early molecular response
5. Five years after initiation of the first BCR-ABL TKI, AA is admitted to the hospital for an unrelated condition. His peripheral blood work shows elevated WBCs and platelets. His BCR-ABL transcript level is 105% and mutation analysis reveals T315I mutation. What is the most appropriate agent for this mutation?
a) Ponatinib
b) Imatinib
c) Dasatinib
6. AA presents to the pharmacy and enquires about OTC supplement St John’s wort. You are at data entry, and notice AA is on a BCR-ABL TKI. What are your next steps?
a) Encourage him to buy a small quantity of the lowest dose in case it doesn’t work
b) Inform him St. John’s wort interacts with his metformin and suggest SAM-e instead
c) Tell him NOT to take St. John’s wort and refer him for assessment for depression
7. What type of resistance is AA exhibiting?
a) Primary resistance
b) Secondary resistance
c) Tertiary resistance
8. BB is a 28-year-old female patient just diagnosed with chronic phase CML. She is eager to start a family. What generation of BCR-ABL TKI should the provider initiate?
a) First generation TKI
b) Second generation TKI
c) Third generation TKI
9. Post pregnancy, BB resumes on BCR-ABL TKI and wants to breast feed the baby. Which of the following statements is TRUE?
a) BB can breastfeed the baby due to formula shortage
b) BB cannot breastfeed the baby as TKIs pass into breast milk
c) BB can breastfeed 6 hours after the TKI dose
10. A patient with chronic phase CML has been previously treated with at least two TKIs. He is unable to tolerate asciminib 20 mg twice a day. What is the next course of action?
a) Reduce dose to 10 mg twice a day
b) Reduce dose to 5 mg twice a day
c) Discontinue the medication
Pharmacy Technician Post Test (for viewing only)
Pharmacy Technician Post-test
Questions
1. A patient recently diagnosed with CML who started on a BCR-ABL TKI presents to the pharmacy counter with a rash on his face. He wants to know if he can use over the counter hydrocortisone cream. What is your next step?
a) Direct him to the hydrocortisone cream in the OTC aisle
b) Inform the patient the rash will disappear in a few days
c) Alert the pharmacist for counseling.
2. In which stage of CML do patients present with at least 20% blasts in the peripheral blood?
a) Chronic Phase
b) Accelerated Phase
c) Blast Phase
3. Which one of the following is a first generation TKI?
a) Dasatinib
b) Nilotinib
c) Imatinib
4. Which BCR-ABL TKI is most likely to cause pleural effusion as an adverse effect?
a) Dasatinib
b) Nilotinib
c) Imatinib
5. If quantitative PCR test shows BCR-ABL transcripts at 3.2%. What type of response is the patient exhibiting?
a) Complete molecular response
b) Major molecular response
c) Early molecular response
6. Which TKI is active against T315I mutation?
a) Ponatinib
b) Imatinib
c) Dasatinib
7. Which of the following patients would you refer to the pharmacist?
a) A patient who is on a BCR-ABL TKI and says her BCL-ABR is below 0.1%
b) A patient who was on a BCR-ABL TKI in 2020 and is now pregnant
c) A patient who is on a BCR-ABL TKI and asks for a bottle of St. John’s wort
8. Which of the following BCR-ABL TKI does not interact with over-the-counter antacids or proton pump inhibitors?
a) Bosutinib
b) Imatinib
c) Nilotinib
9. BB is a 28-year-old female patient just diagnosed with chronic phase CML. She plans to start a family as soon as the oncologist gives her the go-ahead. Which generation of TKI is most appropriate for BB?
a) First generation TKI
b) Second generation TKI
c) Third generation TKI
10. A patient on dasatinib presents to the pharmacy with cough, congestion, and chest pain. He wants to know which aisle the OTC cough and cold products are in. What should you do next?
a) Direct him to the cough and cold aisle
b) Alert the pharmacist for counseling
c) Counsel the patient to discontinue dasatinib
References
Full List of References
References
1. How a CML clinical trial and Gleevec saved my life. Accessed July 1, 2022. https://www.mdanderson.org/cancerwise/how-a-cml-clinical-trial-and-gleevec-saved-my-life.h00-158912490.html
2. CML Patient Story: From Terminal Cancer to 26-Year Survivor. Accessed July 1, 2022. CML Patient Story: From Terminal Cancer to 26-Year Survivor | Patient Power
3. Longo DL. Imatinib Changed Everything. N Engl J Med. 2017;376(10):982-983
4. Vener C, Banzi R, Ambrogi F, et al. First-line imatinib vs second- and third-generation TKIs for chronic-phase CML: a systematic review and meta-analysis. Blood Adv. 2020;4(12):2723-2735
5. Jabbour E, Kantarjian H. Chronic Myeloid Leukemia: 2022 Update on Diagnosis, Therapy and Monitoring [published online ahead of print, 2022 Jun 25]. Am J Hematol. 2022;10.1002/ajh.26642
6. Haider MZ, Anwer F. Genetics, Philadelphia Chromosome. [Updated 2021 Jul 21]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022. Accessed July 9, 2022. https://www.ncbi.nlm.nih.gov/books/NBK560689/
7. Rizzieri D, Moore JO. Implementation of management guidelines for chronic myeloid leukemia: perspectives in the United States. P T. 2012;37(11):640-648.
8. Eden RE, Coviello JM. Chronic Myelogenous Leukemia. [Updated 2022 May 13]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022. Accessed July 9, 2022. https://www.ncbi.nlm.nih.gov/books/NBK531459/
9. Akwaa F, Liesveld J. Surrogate end points for long-term outcomes in chronic myeloid leukemia. Leuk Lymphoma. 2013;54(10):2103-2111.
10. National Comprehensive Cancer Network. Chronic Myeloid Leukemia (Version 3. 2022) cml.pdf (nccn.org)
11. Schiffer AC, Atallah E. Initial treatment of chronic myeloid leukemia in chronic phase. In: Rosmarin GA, ed. UpToDate. UpToDate; 2020. Accessed July 1, 2022. Initial treatment of chronic myeloid leukemia in chronic phase - UpToDate
12. Gleevec [package insert]. Novartis Pharmaceuticals Corporation; 2008
13. Etienne G, Cony-Makhoul P, Mahon FX. Imatinib mesylate and gray hair. N Engl J Med. 2002;347(6):446.
14. Reff MJ, Shillingburg A, Shah B, Elder C, Prescott H, Kennerly-Shah J. Front-line use of tyrosine kinase inhibitors in chronic phase chronic myeloid leukemia: Practice considerations. J Oncol Pharm Pract. 2020;26(1):156-174.
15. Tasigna [package insert]. Novartis Pharmaceuticals Corporation; 2010
16. Hochhaus A, Saglio G, Hughes TP, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016;30(5):1044-1054
17. Sprycel [package insert]. Bristol-Myers Squibb Company; 2006
18. Amrein PC. Low-dose dasatinib: when less can be more. Lancet Haematol. 2021;8(12):e867-e868.
19. Bosulif [package insert]. Pfizer Labs; 2017
20. Patel AB, O'Hare T, Deininger MW. Mechanisms of Resistance to ABL Kinase Inhibition in Chronic Myeloid Leukemia and the Development of Next Generation ABL Kinase Inhibitors. Hematol Oncol Clin North Am. 2017;31(4):589-612.
21. Koji S, Jabbour E, Issa CG, et al. Outcomes of Patients with Chronic Myeloid Leukemia Treated with Third-Line Tyrosine Kinase Inhibitors. Blood 2020; 136 (Supplement 1): 25–26.
22. Iclusig [package insert]. Takeda Pharmaceuticals America Inc; 2022
23. Cortes JE, Kim DW, Pinilla-Ibarz J, et al. Ponatinib efficacy and safety in Philadelphia chromosome-positive leukemia: final 5-year results of the phase 2 PACE trial. Blood. 2018;132(4):393-404.
24. FDA Drug Safety Communication: FDA requires multiple new safety measures for leukemia drug Iclusig; company expected to resume marketing. Accessed July 4, 2022. FDA Drug Safety Communication: FDA requires multiple new safety measures for leukemia drug Iclusig; company expected to resume marketing | FDA
25. FDA approves asciminib for Philadelphia chromosome-positive chronic myeloid leukemia. Accessed July 4, 2022. FDA approves asciminib for Philadelphia chromosome-positive chronic myeloid leukemia | FDA
26. Scemblix [package insert]. Novartis Pharmaceuticals; 2021
27. Cortes, J. E. et al. Asciminib, a first-in-class STAMP Inhibitor, provides durable molecular response in patients (pts) with chronic myeloid leukemia (CML) harboring the T315I mutation: primary efficacy and safety results from a phase 1 trial. Blood 136(Suppl 1), 47–50 (2020).
28. Global Chronic Myeloid Leukemia Pipeline Insight | Clinical Trials Evaluation Research Report 2022 by DelveInsight. Accessed July 4, 2022. Global Chronic Myeloid Leukemia Pipeline Insight | Clinical (globenewswire.com)
29. Di Tullio F, Mandel VD, Scotti R, Padalino C, Pellacani G. Imatinib-induced diffuse hyperpigmentation of the oral mucosa, the skin, and the nails in a patient affected by chronic myeloid leukemia: report of a case and review of the literature. Int J Dermatol. 2018 Jul;57(7):784-790.