Learning Objectives
After completing this application-based continuing education activity, pharmacists and pharmacy technicians will be able to
| • Describe natural kratom products and related chemicals |
| • Differentiate the effects of naturally derived kratom products and those associated with 7-OH-MG or mitragynine pseudoindoxyl |
| • List points of significance important to educating others about products related to kratom |

Release Date:
Release Date: March 15, 2025
Expiration Date: March 15, 2028
Course Fee
Pharmacist: $7
Pharmacy Technician: $4
ACPE UANs
Pharmacist: 0009-0000-25-004-H03-P
Pharmacy Technician: 0009-0000-25-004-H03-T
Session Codes
Pharmacist: 25YC04-VKA34
Pharmacy Technician: 25YC04-AKV68
Accreditation Hours
2.0 hours of CE
Accreditation Statements
| The University of Connecticut School of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education. Statements of credit for the online activity ACPE UAN 0009-0000-25-004-H03-P/T will be awarded when the post test and evaluation have been completed and passed with a 70% or better. Your CE credits will be uploaded to your CPE monitor profile within 2 weeks of completion of the program. |  |
Disclosure of Discussions of Off-label and Investigational Drug Use
The material presented here does not necessarily reflect the views of The University of Connecticut School of Pharmacy or its co-sponsor affiliates. These materials may discuss uses and dosages for therapeutic products, processes, procedures and inferred diagnoses that have not been approved by the United States Food and Drug Administration. A qualified health care professional should be consulted before using any therapeutic product discussed. All readers and continuing education participants should verify all information and data before treating patients or employing any therapies described in this continuing education activity.
Faculty
Amy Nieto PharmD Candidate 2026
UConn School of Pharmacy
Storrs, CT
Michael White PharmD, FCP, FCCP, FASHP
Distinguished Professor and Chair, Pharmacy Practice
UConn School of Pharmacy
Storrs, CT
Jeannette Y. Wick, RPh, FBA, FASCP
Director, Office of Pharmacy Professional Development
UConn School of Pharmacy
Storrs, CT
Faculty Disclosure
In accordance with the Accreditation Council for Pharmacy Education (ACPE) Criteria for Quality and Interpretive Guidelines, The University of Connecticut School of Pharmacy requires that faculty disclose any relationship that the faculty may have with commercial entities whose products or services may be mentioned in the activity.
Dr. White and Ms. Wick and Nieto have no relationship with ineligible companies and therefore have nothing to disclose.
ABSTRACT
People native to Southeast Asia have a deep-rooted history of using kratom, derived from the whole fresh or dried leaves of tropical Mitragyna speciosa trees, dating back centuries. In recent years, kratom-containing products have garnered popularity within the recreational drug market. Consumers can purchase these products in smoke shops, convenience stores, gas stations, and over the Internet without a prescription. Kratom remains a Drug Enforcement Administration (DEA) substance of concern but is not currently scheduled. Notable changes have occurred in the kratom product marketplace. Many products now contain high concentrations of a metabolite 7-hydroxymitragynine (7-OH-MG) or its metabolite mitragynine pseudoindoxyl. Kratom and related compounds work at the μ-opioid receptors to varting degrees. The synthetic products are stronger and more likely to cause misadventure and dependence.
CONTENT
Content
INTRODUCTION
Kratom. What a funny little word. It's six letters and preponderance of consonants give it an explosive, almost exotic sound. When Pete, a 31-year-old construction worker who has been on workers compensation for six months for a back injury asks about it, the pharmacist, Cindy, is baffled. Educated to never respond with “I don’t know,” Cindy replies, “I don’t know but I will gather some information. Can you give me a couple of days to do some research?” Pete agrees and Cindy gathers information using open-ended questions to determine his goals. Pete says he heard that kratom relieves pain much like opioids do, and it’s a “natural product.” He says that his physician is weaning him from opioids, but he still has pain unrelieved by nonopioid pain killers. His bowling league teammate said “7” would be “better than that regular kratom stuff.” He sees both products at the gas station, and he would like to know if they have the same side effects as opioids do. He says, “I would really like to be done with the constipation.”
PAUSE AND PONDER: What things should Cindy think about as she assesses Pete’s situation?
People native to Southeast Asia (Cambodia, Indonesia, Malaysia, Myanmar, Papua New Guinea, and Thailand) have a deep-rooted history of using kratom, derived from the whole fresh or dried leaves of tropical Mitragyna speciosa trees, dating back centuries.1, 2 Once harvested, the leaves can be chewed or brewed into a tea for multiple purposes. Western medical literatures’ earliest mention of kratom dates to the 19th Century. Traditional uses of kratom in Southeast Asia include improving productivity and combatting physical fatigue in manual laborers, treating morphine dependence, and enhancing religious ceremonies.3
In recent years, kratom-containing products have garnered popularity within the recreational drug market. Approximately five million Americans (aged 12 years old or older) have used kratom.4 The predominant demographic of users has recently shifted from middle-aged males to younger people and females.5 Consumers can purchase these products in smoke shops, convenience stores, gas stations, and over the Internet without a prescription.4 In the United States (U.S.), kratom is available in several dosage forms (capsules, concentrated extracts, powder, raw leaves, and tablets) and doses (depends on the dosage form). Around the world, traditional kratom products have been used as a replacement for traditional opioids or other substances of abuse, or for treatment of anxiety or depression. However, no clinical trials substantiate these benefits.6,7,8
Cindy focuses on the allegation that kratom and similar products are “natural,” recalling that “natural” is not equivalent to “harmless.” Natural products are often contaminated with elemental impurities (also known as heavy metals).9 Kratom specifically can be tainted with heavy metals like arsenic, lead, manganese, and nickel. The Food and Drug Administration (FDA) specifies the permissible daily exposure (maximum acceptable intake) for arsenic, lead, and nickel in dietary supplement products and the tolerable upper intake level of manganese would also be exceeded by some products. However, some kratom products have been found to exceed these regulatory thresholds; non-extract products (capsules, powders, and tablets) often have higher concentrations compared to extracts.5
A potential cause of lead and nickel contamination is the country of origin of the kratom trees (Indonesia and Thailand, for example, naturally have greater lead concentrations in their soil and water).10,11 Local water is used to wash the picked leaves which may leave an elemental impurity residue and the trees themselves can pick up the heavy metal from the soil. Machinery use (harvesting, grinding, storing, and shipping), pesticide and herbicide use, and volcanic rock leeching are the ultimate sources of the heavy metals.10,11 Regardless of the source, elemental impurities may be retained on harvested, dried leaves which can then be sold to consumers leading to excessive heavy metal exposure. Some products sold as kratom in the U.S. have been adulterated with synthetic compounds such as 7-hydroxymitragynine ((7-OH-MG), an alkaloid of kratom and a growing concern nationally) or even with fentanyl or hydrocodone.12
Readers should take a moment to review the various chemical names and pronunciations used in this continuing education (CE) activity in the SIDEBAR.
SIDEBAR: Chemical Components of Kratom
- Alkaloids: natural organic compounds that provide varying physiological effects on animals
- Corynantheidine*: a minor alkaloid of kratom with antinociceptive activity
- Corynoxine/corynoxine B: a minor alkaloid of kratom and derivative of corynantheidine; full agonist of the μ-opioid receptor
- Mitragynine*: a major (most abundant) alkaloid of kratom; provides pain-relieving and stimulating effects
- Mitragynine pseudoindoxyl: a 7-OH-MG metabolite; a more potent μ-opioid receptor agonist compared to mitragynine
- Paynantheine: a major, secondary alkaloid of kratom with high binding affinity to serotonin receptors (5-HT1A and 5-HT2B)
- Speciofoline: a minor alkaloid of kratom with no measurable binding affinity at the µ-, δ- or ƙ-opioid receptors
- Speciogynine: a minor alkaloid of kratom with high binding affinity to serotonin receptors (5-HT1A and 5-HT2B)
- Specioliatine*: a minor, rare alkaloid of kratom; modulates the pain-relieving and stimulating effects of mitragynine
- 7-hydroxymitragynine (7-OH-MG): an active metabolite of mitragynine; a potent partial μ-opioid receptor agonist exhibiting pain-relief, sedation, and opioid-induced addiction potential (as indicated in animal studies; see below)
*partial agonist of the μ-opioid receptor
Kratom remains a Drug Enforcement Administration (DEA) substance of concern but is not currently scheduled.6 Kratom products’ overall risk profile is much lower than some substances of abuse (traditional opioids, amphetamines, cocaine) and similar or slightly lower than cannabis and alcohol.7 However, when traditional kratom products are used to treat substance use disorders (SUDs), two observational studies (N = 160, N = 32, respectively) found fewer subsequent episodes of illicit substance abuse and fewer risky lifestyle behaviors such as injecting drugs and needle sharing.9,13
Another observational study (N = 163) found a lower likelihood of constipation, cravings, fatigue, insomnia, and sexual performance issues than those associated with traditional opioid use.14 One small clinical trial (N = 26) demonstrated that traditional kratom products can increase pain tolerance. Following a cold pressor task (a procedure where participants immerse their dominant hand into an ice bath and verbally report pain onset), kratom reduced pain sensitivity. The study also suggests that chronic use may cause hyperalgesia (an increased sensitivity to pain) when kratom alkaloid concentrations are low compared to drug-naïve states.6,15 Whole leaf kratom has modest stimulant effects, so the risk of respiratory depression is low but it does produce adverse events, especially in higher doses, and it has the potential to cause a kratom use disorder (kratom-specific substance use disorder, K-SUD).6,16 The main adverse events include nausea and vomiting, constipation, tachycardia, hypertension, agitation, confusion, elevated liver function tests, and seizures.
Notable changes have occurred in the kratom product marketplace. In January 2024, an exposition (trade show) in Las Vegas for buyers and sellers of legal recreational substances hosted just two companies selling products with high concentrations of a metabolite (7-OH-MG) as the most abundant alkaloid.17 By the time the next exposition was held in Chicago in June 2024, 37 companies in the marketplace with 7-OH-MG as a prominent ingredient in a product or products attended; it’s likely that there are many more that did not attend.17 According to the National Drug Early Warning System (surveillance system that detects early signals of potential drug epidemics), chatter about 7-OH-MG had increased steeply. It rose from roughly 10 posts weekly between September 2023 to November 2024 and spiked to about 20 posts for several weeks from November 2023 to March 2024. From March to June 2024, posts spiked considerably to 30 to 45 posts weekly for several weeks.18 This CE activity will attempt to tackle several key questions regarding this trend towards higher 7-OH-MG products including
- How does the alkaloid composition in traditional kratom products compare with high 7-OH-MG concentration products?
- What do preclinical studies (studies conducted in animal models) suggest about the pharmacologic effects of traditional kratom products compared to high 7-OH-MG concentration products?
- What insight can be gleaned from consumers about the comparative effects of taking traditional kratom products rather than high 7-OH-MG products?
TRADITIONAL KRATOM VS. 7-HYDROXYMITRAGYNINE
As displayed in Figure 1, the most abundant alkaloid in traditional kratom products is mitragynine, with concentrations ranging from 54% to 66% of the total alkaloid content.1 Many other alkaloids comprise the remaining 34% to 46% of total alkaloids, but 7-OH-MG only constitutes less than 1% of the total.
Figure 1. Histogram of the Relative Percentage of Alkaloids in Traditional Kratom Products Versus 7-Hydroxymitragynine Products
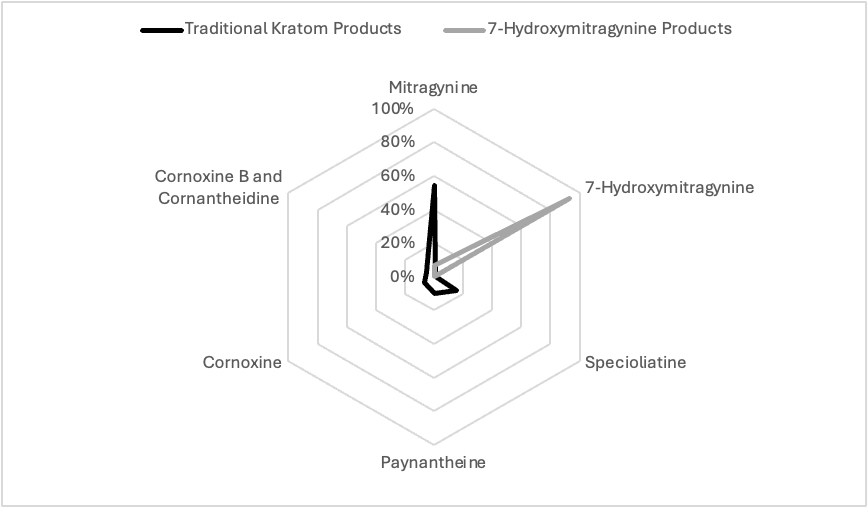
READING THIS HISTOGRAM: Whole leaf kratom (either fresh or leaf) has 54% mitragynine, 16% specioliatine, 12% cornoxine/cornoxine B, 10% paynantheine, and <1% 7-hydroxymitragynine. 7-OH-MG products contain 93% 7-hydroxymitragynine, 7% mitragynine, and none of these other natural alkaloids.
In its natural, fresh state, kratom leaves do not contain 7-OH-MG; however, 7-OH-MG is a degradation product found minutely in older leaves.1 In one laboratory evaluation of 53 commercial kratom products, mitragynine, and 7-OH-MG concentrations were related to the speciofoline concentration, a major alkaloid found in the kratom plant that has unspecified activity at the mu and kappa opioid receptors.19 “High speciofoline” varieties comprised 4.13 mg/G of kratom powder with mitragynine and 7-OH-MG content of 8.13 mg/G and 0.03 mg/G, respectively. “Low speciofoline” varieties had undetectable speciofoline content but mitragynine and 7-OH-MG content of 11.45 mg/G and 0.06 mg/G, respectively. While not found in kratom leaves, the active metabolite of 7-OH-MG is an alkaloid called mitragynine pseudoindoxyl.19
Although several brands of kratom are available, a popular product is called 7-OHMZ. In this product’s certificate of analysis (CoA; see SIDEBAR), the manufacturer reports 14.9 mg of 7-OH-MG and 1.3 mg of mitragynine per tablet (92% 7-OH-MG).20 A different product, Hydroxie, is an ultra-pure 7-hydroxy kratom. Its CoA shows it contains 16.5 mg of 7-OH-MG and 1.3 mg of mitragynine (93% 7-OH-MG).21 A third product, Press’d, has an available CoA that indicates it contains 18.5 mg of 7-OH-MG and 1.5 mg of mitragynine (93% 7-OH-MG).22 The 7-OHMZ and Hydroxie companies state they extract the 7-OH-MG naturally for their products. However, high speciofoline dried kratom leaves contain only 0.03 mg/G of 7-OH-MG. To make a single 7-OH-MG dose with traditional kratom powder, 7-OHMZ and Hydroxie requires between 466 g to 550 g of dried kratom leaves and Press’d requires 616 g of dried leaves which is an entire tree’s harvest of leaves.19
SIDEBAR: TECH TALK: Understanding Product Verification Absent FDA Approval23
As kratom and 7-OH-MG products become more available, consumers need to be aware that they are not FDA-approved. Without FDA approval, the FDA takes no responsibility for ensuring that the kratom or 7-OH-MG concentration on the label matches the product content. Monitoring these products is especially important.
Plants can absorb heavy metals, pesticides, and other potentially harmful chemicals that may be in the soil or water easily, and analysts find these contaminants in the leaves, flowers, and stems. Therefore, consumers—and the pharmacists and technicians who advise them—need to investigate the kratom products available on the market if they make recommendations or find a product is widely used in their localities.
Consumers can request products’ Certificate of Analysis (CoA), which provides information about testing for contaminants and active ingredient levels. If the retailer cannot provide it, consumers should avoid that product. States and retailers are starting to take the initiative to ensure consumers have needed information.
When looking at the CoA, consumers can be more confident of the quality if the CoA states that the lab meets “ISO 17025” standards. Consumers can also look to see if the CoA states that the lab complied with the standards set by one of three organizations:
- the Association of Official Agricultural Chemists (AOAC),
- the American Herbal Pharmacopoeia (AHP), or
- the U.S. Pharmacopeia (USP).
Consumers should beware of products that list the total concentration in the product (i.e., 250 mg in the bottle) and not the active ingredient concentration (i.e., 2.5 mg/mL). Of course, products should clearly define a “dose,” and list the amount of active ingredient in a dose and not in the entire product. These products may contain other related compounds besides mitragynine and 7-OH-MG. Consumers should also beware of “proprietary blends” where the alkaloids in the product are given with a total dosage but the amount of each alkaloid is not provided.
PHARMACOLOGIC COMPARISON OF ALKALOID PHARMACOLOGY
Results of In Vitro Studies
Stimulating the μ-opioid receptor with traditional opioids (like morphine) has been associated with feelings of wellbeing, euphoria, pain relief/antinociception (reduced sensitivity to pain), and constipation. It can also lead to central nervous system depression that impairs the normal respiratory response to rising carbon dioxide levels in the blood. Mitragynine and 7-OH-MG are partial μ-opioid agonists while morphine is a full agonist.1 In an in vitro study, the μ-opioid receptor Ki receptor binding affinity (lower Ki means tighter binding) and EC50 for cAMP accumulation (lower cAMP accumulation means greater suppression of adenylate cyclase production) were determined for mitragynine and 7-OH-MG. The μ-opioid receptor Ki for mitragynine was 15-fold higher than 7-OH-MG (238 nM vs. 16 nM) and 159 times higher than morphine (238 nM vs. 1.5 nM). The μ-opioid receptor cAMP EC50 was 29-fold (398 nM vs. 13.6) higher for mitragynine versus 7-OH-MG and 83-fold (398 nM vs. 4.8 nM) higher than morphine.19 In other words, 7-OH-MG and morphine bind tighter to the μ-opioid receptor and suppress adenylate cyclase production at magnitudes far greater than mitragynine. For reasons that are poorly understood, mitragynine and 7-OH-MG are less likely to cause respiratory depression than traditional opioids in preclinical studies.6,19 Other kratom alkaloids (specioliatine, paynantheine, and speciogynine) bind weakly to μ-opioid receptors with Ki values ranging from 410 to 728 nM.19
Understanding how these products work and their effects on various systems is emerging. The composition of kratom is highly variable; although mitragynine is the predominant alkaloid, around one-third of kratom is composed of other, minor alkaloids. Whether minor or major, kratom’s alkaloids have specific binding affinities and therapeutic outcomes. Physiologic effects of kratom therefore depend on all the alkaloids present. For example,
- Stimulating alpha-1 and alpha-2 adrenoceptors with mitragynine provides modest antinociception (pain reduction). These effects can be attenuated by alpha-1 and alpha-2 antagonists (prazosin—an antihypertensive—and yohimbine—a supplement allegedly used for benign prostatic hyperplasia and sexual function, respectively).19
- Stimulating alpha-2 adrenoceptors, like with clonidine, can prevent sympathetic outflow (signaling leading to activation of the sympathetic nervous system) in people with withdrawal symptoms from traditional opioids. Research indicates this effect may attenuate opioid withdrawal symptoms that patents experience.
- Mitragynine has moderate nonselective agonist effects across alpha-1 and alpha-2 adrenoceptors with Ki values ranging from 1.3 to 9.3 mcM.
- Speciogynine provides strong alpha-2 agonist effects for many alpha-2 adrenoceptors (Ki for alpha-2A, alpha-2B, and alpha-2C from 0.4 to 2.6 mcM) but has no effect on alpha-1 adrenoceptors.
- Corynantheidine is a potent agonist for alpha-1D adrenoceptors with a Ki value of 0.042 mcM. Stimulation of serotonin receptors can enhance mood and alertness.
- Mitragynine has modest agonist effects at serotonin receptors with Ki values at 5-HT-1A, -2A, and -2B ranging from 1.3 to 5.9 mcM.
- Paynantheine and speciogynine are potent agonists at serotonin receptors with Ki values for 5-HT-1A and -2B ranging from 0.02 to 0.04 mcM and 5-HT-2B Ki values ranging from 0.8 to 1.3 mcM.19
Traditional kratom products’ pharmacology is complex. Consider this: the human enzyme CYP3A4 converts a portion of mitragynine to 7-OH-MG in the brush border of the gastrointestinal tract and the liver. Next, CYP3A4 circulating in the plasma converts a portion of this active metabolite to mitragynine pseudoindoxyl, a molecule with the ability to interact with central opioid receptors.24 Mitragynine pseudoindoxyl is at least as potent an opioid agonist as 7-OH-MG and lacks alpha adrenoceptor or serotonin receptor effects.25
Human studies are needed to explore kratom's utility in analgesia, euphoria, and addiction more thoroughly. As of January 10, 2025, only nine ongoing studies were listed in clinicaltrials.gov. Most were small, although a few larger studies are underway.26,27 In one now complete study, human plasma converted 7-OH-MG into mitragynine pseudoindoxyl much more effectively than plasma from other species.25 The percentages of mitragynine pseudoindoxyl formed at the end of 120 minutes of incubation with 7-OH-MG in dog, monkey, mouse, and rat plasma were below 4.3% but 53.8% in human plasma. This suggests that mitragynine pseudoindoxyl’s contribution to the overall opioid receptor’s impact could differ between species. The impact is that pharmacologic effects might also differ between animals and humans with more dangerous effects in people.25 Taken together, the receptor pharmacology of traditional kratom products is a constellation of μ-opioid, alpha-1 and alpha-2 adrenergic, and serotonin receptor effects from multiple alkaloids while 7-OH-MG products focus almost exclusively on μ-opioid receptor effects, which is similar to traditional opioids.1,6,19,24,25
PAUSE AND PONDER: What takeaways can you list for the heavy science presented in the last few paragraphs?
As Cindy assembles all of the information, she is somewhat confused. She calls a professor at her alma mater and asks if he can discuss these findings with her. After an hour of discussion, she says, “Let me summarize what I have learned so I can explain this to my patient:
- Kratom alkaloids in the highest abundance in whole leaf products primarily interact with alpha-1 and alpha-2 adrenergic and serotonin receptors with indirect and weak opioid effects
- Metabolism of mitragynine via CYP3A4 results in metabolites (7-OH-MG and then mitragynine pseudoindoxyl) that more potently exhibit opioid agonist properties
- 7-OH-MG binds the μ-opioid receptor far greater than mitragynine and 7-OH-MG products have far more 7-OH-MG than whole leaf products could ever provide
- Differences in metabolite amounts and μ-opioid receptor quantities and locations may be species-specific but the effects seen with 7-OH-MG in animals would likely be greater in humans.”
Cindy’s professor says that she got it! He suggests that now that she knows the basic pharmacology, she should move on to some animal studies.
Results of Animal Studies
A step up from in vitro study is the use of animal models. Researchers select animal models carefully based on the purpose of their study. In the case of kratom and 7-OH-MG products, researchers’ intent is to determine how the products compare to opioids in terms of pain relief, euphoria, and addiction potential. For that purpose, the ideal models are small rodents (rats and mice). Many rodent models of pain and addiction exist. Many of the experimental animals can and will self-medicate in response to pain or craving.
A mouse model (tail-flick test) study sought to establish the equipotent antinociception doses of mitragynine and 7-OH-MG.1 Overall, mitragynine 140 mg/kg subcutaneously provided a similar tail-flick response to 0.7 mg/kg of 7-OH-MG. (See how much more potent 7-OH-MG is compared to mitragynine?) The brain concentrations of 7-OH-MG were almost identical in the mitragynine group (with 7-OH-MG created via metabolism) and the group given 7-OH-MG, suggesting that 7-OH-MG is a driver of mitragynine’s opioid mediated pain-relieving effects.1 In another mouse study, 7-OH-MG produced a potent antinociceptive effect, similar to morphine and mainly through activation of μ-opioid receptors.28 Tolerance to 7-OH-MG’s antinociceptive effect developed, similar to the tolerance that develops with morphine. Importantly, cross-tolerance to morphine was evident in mice rendered tolerant to 7-OH-MG or morphine when the drugs were switched. Finally, naloxone-induced withdrawal signs occurred equally in mice chronically treated with 7-OH-MG or morphine.28
Overall, 7-OH-MG appears to have greater abuse potential than mitragynine.29 In a rat model, investigators took morphine-addicted rats and allowed them to self-administer 7-OH-MG or mitragynine in a substitution phase. Rats preferred 7-OH-MG over mitragynine and did so in a manner that was dose-dependent and similar to their previous morphine use. After the substitution phase, the researchers allowed morphine self-administration again. Rats using 7-OH-MG ingested higher morphine doses than they had previously while those who used mitragynine ingested lower morphine doses. Nalxonaxine, an experimental μ-opioid receptor antagonist, reduced 7-OH-MG and morphine self-administration doses.29
Less is known about mitragynine pseudoindoxyl’s antinociceptive and withdrawal effects.30 In one model, researchers tested mice to see how long they could retain their tails in a hot water bath at baseline. Then they administered repeated saline or escalating doses of morphine, kratom alkaloid extract, mitragynine, or mitragynine pseudoindoxyl for four days. Immediately before day 5 dosing, the mice previously treated with morphine (~11 + 2 to 2 + 1 s), kratom extract (~10 + 1 to 4 + 1 s) or mitragynine (10 + 1 to 4 + 1 s) demonstrated significantly more drug-induced hyperalgesia, but the normal saline group did not (~10 + 1 to 8 + 1 s). The mitragynine pseudoindoxyl group had reductions from baseline (~11 + 2 to ~6 + 1 s) but this did not achieve statistical significance. Mice were then administered naloxone on day 5 after their assigned drug was administered. When exposed to naloxone, all active groups had less severe withdrawal symptoms than the morphine group for some measures, although for some measures the withdrawal symptoms were greater with the kratom extract, mitragynine, and mitragynine pseudoindoxyl groups than the normal saline group.30
These studies suggest that 7-OH-MG has more potent antinociceptive effects, opioid users would be more likely to seek it, and would promote the subsequent use of higher doses of traditional opioids (called a reinforcing effect) than mitragynine.28,29,30
In a recent editorial, prominent kratom basic scientists expressed grave concern about 7-OH-MG products.31 They believe that manufacturers are taking mitragynine extract and chemically converting it into 7-OH-MG producing unknown alkaloids in the process. Creating alkaloids that do not exist in nature or as a known metabolite in the human body poses unknown risks to patients. They also believe that the pharmacology of 7-OH-MG is inherently riskier than that of mitragynine.31
PAUSE AND PONDER: What is the pharmacist’s role in preventing kratom misuse? How would you advise your patients to approach using this product safely?
Kratom this. Kratom that. Cindy is learning all about kratom extracts! After reviewing the animal studies, she calls her professor back. Concerned with the opioid-like properties of kratom’s alkaloids and metabolites, Cindy is unclear if kratom would be a good alternative for Pete’s pain. Cindy’s professor, understanding her concern, sends her to search the World Wide Web for personal, raw experiences from users.
ANECDOTAL EFFECTS OF 7-HYDROXYMITRAGYNINE PRODUCTS
Anecdotal experiences with kratom include discussions of pain relief, energizing or relaxing effects, or its use to prevent opioid withdrawal in between opioid doses, or as a chronic replacement for opioids.6,8,9,13,14 The euphoric effects caused by traditional opioids reportedly do not occur for many consumers who take traditional kratom products; this makes traditional kratom products an unsuitable substitute for people who use them recreationally.8 There is said to be a ceiling effect for the mood elevation with traditional kratom products and that nausea experienced when escalating doses tempers the desire to binge dose.6,32
The reported consumer experiences with 7-OH-MG products to date are characteristically different than experiences users describe with traditional kratom products.32 Table 1 provides some direct quotes from Reddit chat rooms concerning 7-OH-MG. They are categorized into initial experiences, addiction and withdrawal, and the use of kratom to manage 7-OH-MG withdrawal.32 To summarize these comments, users described 7-OH-MG products’ euphoric effects as more intense. Although the users didn’t report if the pain was due to injury or to hyperalgesia. They reported pain relief but indicated that effects were short lived. 7-OH-MG products can lead to rapid dose increases, with tolerance and withdrawal developing more quickly than with traditional kratom products. A 7-OH-MG addiction can result in financial distress as consumers lose the ability to control their use. A three-tablet pack of Press’d 7-OH-MG 18 mg costs $17 and the suggested dose is ½ tablet. If patients take ½ tablet twice a day, that is $5.70 per day or $2,081 per year. Extrapolating to recreational use and tolerance, patients who take 12 tablets a day expend $68 a day or $24,820 a year. Confusing 7-OH-MG products with traditional kratom products can lead to relapse for consumers using kratom as a chronic opioid substitute.
Table 1. Anecdotal customer experiences with 7-hydroxymitragynine products.32
| Early Experiences | · “…a lot of people are strictly using 7oh for the biggest burst of high they can get. People will tiptoe around it saying they want pain relief but if the euphoria burst isn't there, they don't want it.”
· “Some kratom is like a beer, with 7oh is like a shot.” · “…7oh doesn't have that ceiling effect of nausea the same way kratom does…Proceed with caution though 7oh is a different animal.” · “the cleanest and more euphoric high that I’ve gotten from… any kratom shots or powders.” · “kratom has a pretty low ceiling for effects but with 7 hydroxy you can get rlly [sic] up there without getting nauseous or wobbly.” · “7oh makes me more anxious and can even cause some very severe anxiety symptoms and racing thoughts.” “I personally would actively avoid 7oh if I didn’t have my anxiety under control.” · “I get some rebound anxiety when the 7OH dose wears off, usually like 1.5-2 hours after taking it. I’d bet the higher the dose the worse that becomes.” · “Be really careful with 7oh.” · “Put it under my tongue and at first I felt underwhelmed. But about 25 minutes in I felt this sudden, powerful buzzing numbness flush my body. This lasted for about an hour and then I kept drifting to and from sleep+dreams all cozey on the couch. I was super itchy and not exactly comfortable for the first while but the later hours made up for that. Will do again, sparingly.” · “It’s a different feeling to start with, itchier and more intense. Like your whole body’s blushing. And although ive got kratom nods before, they were not as couch-lock-ey or as long as this was.” |
| Addiction and Withdrawal | · “I am not sure how to proceed. Ugh, it's like I am right back on the oxy merry go round after working so hard to get off, and being so happy to be free (with the exception of plain leaf kratom). I am on about 3 tablets a day now. How do we get off? Use massive doses of plain leaf?”
· "I take it everyday 3x a day and I wakeup in withdrawal. It really sucks I feel like a fool getting mixed up in this world. Last time I tried to wean down I got so depressed it was scary. Been using for 6 months. It's like getting hooked on oxy once you enter the everyday use and it's been a few months. It creeps up slowly but it's HARD to get off." · “To give you perspective, I start feeling withdrawals after 4 hours and it is hard for me to go any longer than that . I wish I could make it a full 24 hours. Albeit I do take .5 or 1 tab at a time, but I do it at least 4 times a day or I’m miserable.” · “…will be tianeptine for me over again.” [Referring to the tricyclic drug tianeptine which has potent opioid like effects and was being sold illegally as a dietary supplement in 2024 throughout the country] · “Be careful. Be super careful, I had to go to detox to get off it. The withdrawals are insane.” · “Much worse than kratom. Not as bad as fent or heroin…” · “Anyone else get restless legs/ restless when stopping/ cutting back a lot?” · “Yeah, unfortunately 7oh is physically addictive. I quit the beginning of July and it sucked. It’s too expensive to be so dependent on.” · “...introduced to this stuff about 4 weeks ago and went nuts. Now it's time to say good bye. I can not throw my life away for this stuff.” · “I can already tell stopping is going to be ugly. Anyone got any hints on how to dry out from this stuff.” · “I was taking 8 a day for several months. Have just over 55 hours off now. Tapered down to a half in the morning and a half at night. Had to just be okay with feeling pretty shitty all of the time, felt like crying a lot randomly.” · “I take 3 of these at a time now and do that 3 or 4 times a day. Draining my accts, trying to come off of it because the WDs suck.” · “If I don’t take plain leaf with it I’ll wake up 2x in the night withdrawing. Stuff has a shorter half life than u would think. Atleast for me. It’s physically addicting.” · “I was taking around 8-10 tabs a day. Did this for around 1-2 months. Expensive habit. Tried to just stop several times, only to wake up the next day in severe withdrawals. Would drive myself to the smoke shop sick as shit because I have a job and need to be able to function.” “Motivation is completely drained. I’m very tired and dopamine/serotonin feel completely nonexistent almost. Very hard to get anything done but hopefully things start to improve after a week or two.” “Tightness in my chest. Panic and anxiety. Existential dread. Flu-like feeling across my whole body. Cold and hot flashes. Never warm, always cold, but sweating a lot. Joints hurt. Back hurts. Head hurts. Eyes hurt. No motivation, no drive.” |
| Kratom to Manage 7-Hydroxymitragynine Withdrawal | · “If you're like a lot of people, tapering doesn't really work. I'm not saying it can't be done, I'm just saying it can't for some people. Try regular kratom, which eliminates most the withdrawals pretty well.”
· “Kratom helps with some of the worst of the withdrawals.” · “Now I am just taking 5 capsules in the morning of normal kratom red vein and 5 at night and I still feel kind of sick but I feel immense relief that I’m through the worst of it and no longer driving to the smoke shop sick as shit every couple days and dropping $300-$500.” · “just use regular kratom powder to handle the wds.” · “The anxiety for me is bad before hand but when I jump to leaf its always minimally bad. I feel like a little tired, and anxious because I miss my 7oh babies...” |
Experiencing the euphoric high with 7-OH-MG products (that whole leaf kratom or mitragynine extract products lack) can undo addiction control. Finally, while some consumers try to stop using 7-OH-MG cold turkey, many consumers describe needing to move from 7-OH-MG to whole leaf kratom products or mitragynine extracts to deal with withdrawal symptoms and cravings. Importantly, the posts do not describe people moving from 7-OH-MG to traditional opioids or other illicit opioid-like products such as tianeptine (atypical tricyclic antidepressant with opioid receptor agonist properties; also called “gas station heroin”)(see SIDEBAR). These products were virtually unheard of only a few months ago. It is still too early to know the full measure of adverse events that they could cause.32
SIDEBAR: TIANEPTINE33,34
Globally, several health regulatory agencies have approved tianeptine for the treatment of major depressive disorder since its discovery in the 1980s, but the FDA has not. This molecule, originally called an atypical tricyclic antidepressant, appears to have potential benefits for patients experiencing anxiety and irritable bowel disease, but it does not work like other tricyclics. Later research found it targets the opioid mu receptor. Despite its status as an unapproved drug in the U.S., individuals are able to procure it online and at some retail locations (e.g., bodegas, corner stores, gas stations, mini marts, smoke shops). The origins of these substances is sketchy; it’s possible that they are commercially produced by drug manufacturers but just as likely synthesized in underground laboratories. Tianeptine, labeled as "Zaza" and "Tianna Red," has joined the various gas station drugs and has earned the name “gas station heroin.” It is often packaged in shot-sized bottles, and frequently contains synthetic cannabinoids. Recreational users seek tianeptine for its ability to produce a euphoric, opioid-like high. Sadly, chronic use can lead to dependence, tolerance, addiction, and overdose. Some users have attempted suicide. Calls to poison control centers have skyrocketed since 2014.
WHERE SHOULD WE GO FROM HERE?
Why are these products available for sale legally in the U.S.? As noted, several members of Congress asked the DEA not to list kratom products in Schedule I. Traditional whole leaf kratom products contain a natural ingredient with a blend of alkaloids from dried leaves that have effects on opioid receptors (without reinforcing effects), alpha adrenoceptors, and serotonin receptors.8 Traditional kratom products have been used around the globe for medical purposes for decades and evidence about its relative safety compared to other substances of abuse is increasing.6-9,13,14
Congress's concerns included the fact that a bipartisan majority felt that that ban would be made with no opportunity for public comment from researchers, consumers, and other stakeholders. They indicated that such a precipitous decision could be deleterious to consumer access and choice of an internationally recognized herbal supplement; it could also stop progress on studies targeting the treatment of individuals suffering from opioid and other addictions.35
As Cindy did more research, she found that kratom and related products are illegal in some states even though the federal government has not restricted its use. Restrictions may simply impose age limits, employ a more comprehensive kratom consumer protection restrictions, or may ban these products entirely. The Global Kratom Coalition maintains a map (https://globalkratomcoalition.org/regulation-map)36 with updated information of the restrictions in the U.S. (see reference), but it is always wise to determine the laws in your state from original sources. As of January 1, 2025, many states, including California, Connecticut, Maine, Massachusetts, New Hampshire, New Jersey, New York, and Pennsylvania have no statewide restrictions. Alabama, Arkansas, Indiana, Rhode Island, Vermont, and Wisconsin have outlawed kratom products. Many other states, including Florida, Georgia, and Texas have enacted kratom consumer protection regulations. Healthcare providers need to know if their state has passed bans or regulations, and what the regulations specify. A few localities in states where kratom is legal have established their own restrictions. Some examples include San Diego, California (sale or possession are illegal), Denver, Colorado (no sale for human consumption), Jerseyville, Illinois (banned entirely), Sarasota County, Florida (banned entirely), and Union County, Mississippi (banned entirely).37
The Kratom Consumer Advisory Board is comprised of a scientist and pharmacist (C. Michael White, PharmD) and consumers who have been helped or harmed by kratom products.38 They develop position statements and advise researchers, legislators, and advocacy groups about best practices regarding kratom use. The position statements can be accessed in the references and describe potential regulatory language and rationale behind preventing children from accessing kratom, the risks of 7-OH-MG products, proper labeling, and the need to register products that are being sold to consumers in the state to enforce safety regulations and good manufacturing practices.38
This regulatory environment surrounding kratom should not extend to products with doses of 7-OH-MG that could never exist in nature. Emboldened by the DEA and the FDA’s inaction to date, newer products such as the “Press’d: The Hulk Blend” are combining 15 mg of 7-OH-MG with 15 mg of mitragynine pseudoindoxyl.39 There is one product that contains only mitragynine pseudoindoxyl.40 It is indefensible that these products could possibly be considered natural since mitragynine pseudoindoxyl does not exist in kratom leaves at all. Now, other semi-synthetic alkaloids of mitragynine are being created and placed in products such at 8- and 11-hydroxymitragynine and “Red-OH” with no published animal or human data.41 The HOPES research group at the University of Connecticut is actively cataloging a comprehensive list of 7-OH-MG, mitragynine pseudoindoxyl, 8-hydroxymitragynine, and “Red-OH” products on the market so states that register products sold in their state can assure that these drugs are excluded from sale. If the tianeptine debacle has taught us anything, it is that we need to identify products being sold as natural products and determine if they are truly reflective of nature.42
Finally, new clouds are on the horizon with mitragynine and 7-OH-MG extracts that are being placed into candy formulations (popping crystals, noobs, chocolate bars, gummies, taffy, and ice cream cones), flavorings like strawberry, raspberry, mango, and grape, and sold with cute cartoon mascots.43 The more kratom products resemble sweet treats, the greater the risk of children consuming them. The HOPES research group is also cataloging a comprehensive list of these products that could entice children so states that register products sold in their state can restrict their sales.43
PAUSE AND PONDER: Does your practice state or municipality have regulations towards 7-OH-MG? Mitragynine? Kratom? Should it?
In this CE activity, we have shown that 7-OH-MG and mitragynine pseudoindoxyl relies entirely on opioid receptor effects, rather than impacting a balanced series of receptors like traditional kratom products do.1,8,19,28,29 Preclinical studies suggest that 7-OH-MG has reinforcing effects that mitragynine (the most abundant alkaloid in traditional kratom products) does not have; this is a warning sign for its addiction potential.28,29 Customer experiences with 7-OH-MG products suggest euphoric effects that mimic traditional opioids, faster tolerance and shorter duration of action, and faster onset with more severe withdrawal compared to traditional kratom.32 Until more is known about 7-OH-MG’s effects, shouldn’t customers be protected from accessing products that can potentially result in a lifetime of addiction? If nothing else, can we at least ensure that 7-OH-MG products are not classified as kratom products? If a consumer substitutes kratom for traditional opioids and accidentally experiences the euphoric effects from 7-OH-MG, it might be analogous to giving alcoholics a drink and restarting their cravings. If a wave of bad press about 7-OH-MG products falls under the umbrella of “kratom products,” both products could become Schedule I; a risk to consumers who rely on kratom to manage their opioid use disorder (OUD).
California is working on legislation that would protect their citizens from 7-OH-MG products.43 In its legislation, kratom products would have a dose of 7-OH-MG that is a small fraction of total alkaloids in the product. This proposed California legislation can be a national model or can be emulated state by state.44
What are health professionals’ responsibilities regarding 7-OH-MG products? The first is to be aware of changes in the kratom marketplace. Providers who have patients using traditional kratom products or who inquire about its use need to tell them that 7-OH-MG products—which they may call “7” or “7-OH”—are not kratom and could reinforce opioid addiction rather than provide relief. They should also educate patient’s that the U.S. FDA has approved therapies for OUD where the positive balance of benefits to risks are well known, augmenting that counseling providers can tell patients how to access services.45 If people are addicted to 7-OH-MG products, they need treatment to overcome their K-SUD, just like they would for other opioid receptor agonists. Finally, health professionals should alert regulators and our legislators about emerging threats to our patients’ health and well-being so they can take action.
Cindy finished her research and scheduled an appointment to meet with Pete. She has reviewed all of the science, reconnected with her professor to clarify a few things, and also examined her state and local laws. She looked for articles, podcasts, and radio transcripts to find facts about local, state, and national trends. When Pete arrives, she is prepared to discuss the risks associated with 7-OH-MG. She tells him that “7” is not a natural product, explains why that is the case, and that it could be downright dangerous. Next, she discusses cost (which can be steep) and the fact that it isn’t covered by insurance. She expresses concern that we know little about its safety and gas station products are often labeled poorly or misleadingly. Finally, she tells Pete that it will not “fix” his problem with constipation. Kratom may be a potential option for some patients, but other FDA-approved options are available to support Pete that have proven clinical trial effects and would be better initial options to explore.
Cindy realizes that this is an opportunity to help Pete. She explains that he is having breakthrough pain, and it may be time for the multidisciplinary team to become more involved. Pete admits he has not been going to physical therapy as scheduled, and he blushes when she asks if he’s been taking a stimulant laxative (he hasn’t). Cindy takes some time to contact the primary prescriber, and they work with the physical therapist to develop a better treatment plan for Pete. That new plan does not include 7-OH-MG.
CONCLUSIONS
Products with 7-OH-MG and/or mitragynine pseudoindoxyl act solely through opioid receptors. Preclinical studies and human anecdotal experiences show 7-OH-MG provides euphoric effects that traditional kratom products (whole leaf or mitragynine extracts) do not. Given the known risks of OUD, 7-OH-MG and/or mitragynine pseudoindoxyl products should not be thought of, or listed as kratom and regulators should not transfer the hands-off attitude towards kratom to 7-OH-MG. It is inappropriate to have opioid receptor stimulators like 7-OH-MG or mitragynine pseudoindoxyl sold in gas stations, smoke shops, and over the internet without a prescription, limitations in dosage, and medical oversight.
Pharmacist & Pharmacy Technician Post Test (for viewing only)
Kratom and Knock-offs: Should You Leaf Them Alone?
Post-Test Questions
LEARNING OBJECTIVES
After completing this continuing education activity, pharmacists and pharmacy technicians will be able to
• Describe natural kratom products and related chemicals
• Differentiate the effects of naturally derived kratom products and those associated with 7-OH-MG or mitragynine pseudoindoxyl
• List points of significance important to educating others about products related to kratom
1. Which of the following is the most abundant alkaloid of whole leaf kratom that provides pain-relieving and stimulating effects?
A. Corynoxine/corynoxine B
B. Mitragynine
C. 7-hydroxymitragynine (7-OH-MG)
2. Which of the following is not found in fresh kratom leaves but in miniscule levels in dried kratom leaves?
A. 7-OH-MG
B. mitragynine pseudoindoxyl
C. tianeptine
3. How do 7-OH-MG products differ from traditional whole leaf kratom products?
A. Whole leaf kratom products contain a natural ingredient with a blend of alkaloids from dried leaves that have a range of effects.
B. 7-OH-MG products contain a natural ingredient with a blend of alkaloids from dried leaves that have a range of effects.
C. Both products contain a natural ingredient with a blend of alkaloids added by their manufacturers that have a range of effects.
4. Researchers have found a number of receptors at which traditional kratom products work. Which list is CORRECT?
A. opioid receptors, beta adrenoceptors, and serotonin receptors
B. opioid receptors, alpha adrenoceptors, and norepinephrine receptors
C. opioid receptors, alpha adrenoceptors, and serotonin receptors
5. Two observational studies examined the use of traditional kratom products to treat substance use disorders. What have they found?
A. Kratom provided no benefits whatsoever for SUD.
B. Participants were more likely to return to injecting drugs.
C. Participants were less likely to use illicit substances.
6. Why might traditional kratom products be less likely to cause addiction and elevated recreational use as compared to 7-OH-MG?
A. Traditional kratom seems to have a ceiling effect for mood elevation.
B. Escalating doses causes itching that tempers the desire to binge dose.
C. There is no difference in addiction risk between these products.
7. A patient reports that he is being weaned off opioids and will be using “7” to handle his pain going forward. Which of the following is TRUE?
A. “7” works as well as FDA-approved opioids and it costs significantly less.
B. “7” products could reinforce opioid addiction rather than provide relief.
C. “7” is an excellent alternative to FDA-approved treatments for K-SUD.
8. A patient comes to the pharmacy with a bottle of kratom he bought from a head shop. He says he has listened to a couple of newscasts about kratom and 7-OH-MG products, and he is concerned about heavy metal contamination. Where would you suggest he look for more information?
A. A CoA from an independent lab that complies with recognized quality criteria
B. The Food and Drug Administration-approved product label or insert
C. An Occupational Safety and Health Administration safety data sheet
9. A patient who has been experiencing pain has been on opioids for more than two years. He is considering using 7-OH-MG. When you point out some of its disadvantages, he says that if that information was factual, Congress would have put 7-OH-MG into schedule 1; he has heard that it did not when it reviewed this product. What is the BEST way to address his comment?
A. Congress did not put 7-OH-MG into schedule 1 because doing so could stop studies targeting the treatment of individuals suffering from opioid and other addictions.
B. Members of one political party blocked the scheduling saying it would undermine access to an internationally recognized herbal supplement and limit self-treatment.
C. Congress has not addressed 7-OH-MG; a bipartisan group did ask that kratom NOT be placed in schedule 1 to allow access to this herb and support ongoing studies.
10. After discussing the use of “gas station products” to treat ongoing lingering pain with a patient, the patient decides to continue with FDA-approved products. GOOD JOB! What is the BEST next step to help this patient?
A. Make sure that the patient has not changed her mind about gas station products every single time she comes to the pharmacy to refill an opioid prescription.
B. Make a note in your state’s prescription drug monitoring program that this patient has been asking for illegal opioids and is at high risk for recreational drug use.
C. Determine if the patient’s treatment plan needs adjusting and work with the treatment team to ensure everyone is reinforcing consistent messages.
References
Full List of References
REFERENCES
- Kruegel AC, Uprety R, Grinnell SG, Langreck C, et al.. 7-Hydroxymitragynine Is an Active Metabolite of Mitragynine and a Key Mediator of Its Analgesic Effects. ACS Cent Sci. 2019;5(6):992-1001. doi: 10.1021/acscentsci.9b00141.
- KRATOM (Mitragyna speciosa korth). Drug Enforcement Administration, Diversion Control Division, Drug & Chemical Evaluation Section. April 2024. Accessed January 5, 2025. https://www.deadiversion.usdoj.gov/drug_chem_info/kratom.pdf
- Following “the Roots” of Kratom (Mitragyna speciosa): The Evolution of an Enhancer from a Traditional Use to Increase Work and Productivity in Southeast Asia to a Recreational Psychoactive Drug in Western Countries
- SAMHSA, Center for Behavioral Health Statistics and Quality, National Survey on Drug Use and Health, 2021 and 2022. Accessed January 5, 2025. https://www.samhsa.gov/data/sites/default/files/reports/rpt42728/NSDUHDetailedTabs2022/NSDUHDetailedTabs2022/NSDUHDetTabsSect8pe2022.htm.
- Snow Caroti K, Joseph A, Sapowadia A, Michael White C. Elemental impurities (heavy metals) in kratom products: an assessment of published individual product analyses. Clin Toxicol (Phila). 2024;62(10):651-660. doi:10.1080/15563650.2024.2395552
- McCurdy CR, Sharma A, Smith KE, et al. An update on the clinical pharmacology of kratom: uses, abuse potential, and future considerations. Exp Rev Clin Pharmacol. 2024;17:131-142. doi: 10.1080/17512433.2024.2305798
- Global Kratom Coalition. A Comparative Safety and Risk Analysis. Accessed January 10, 2025. https://static1.squarespace.com/static/6508b3f79033221c2aa1ea17/t/669585eefa4146188d8853a4/1721075184913/Comparative+Safety+of+Kratom+Compared+to+Other+Commonly+Used+Substances+%281%29.pdf
- White CM. Pharmacologic and clinical assessment of kratom: An update. Am J Health Syst Pharm. 2019;76:1915-1925. doi: 10.1093/ajhp/zxz221.
- Singh D, Narayanan S, Abdullah MFIL, et al. Effects of kratom (Mitragyna speciosa Korth.) in reducing risk-behaviors among a small sample of HIV positive opiate users in Malaysia. J Ethn Subst Abuse. 2022;21:1285–1295. doi: 10.1080/15332640.2020. 1845899
- Prozialeck W, Fowler A, Edwards J. Public health implications and possible sources of lead (Pb) as a contaminant of poorly regulated kratom products in the United States. Toxics. 2022;10(7):398. doi: 10.3390/toxics10070398.
- Braley C, Hondrogiannis EM. Differentiation of commercially available Kratom by purported country of origin using inductively coupled plasma-mass spectrometry. J Forensic Sci. 2020;65(2):428–437. doi: 10.1111/1556-4029.14201.
- Grundmann O, Veltri CA, Salari M. Kratom fact sheet for healthcare professionals. March 2019. Accessed January 5, 2025. https://nd.az.gov/sites/default/files/news/Kratom.pdf
- Saref A, Suraya S, Singh D, et al. Self-report data on regular consumption of illicit drugs and HIV risk behaviors after kratom (Mitragyna speciosa Korth.) initiation among illicit drug users in Malaysia. J Psychoactive Drugs. 2020;52:138–144. doi: 10.1080/02791072.2019.168655350.
- Saref A, Suraya S, Singh D, et al. Self-reported prevalence and severity of opioid and kratom (Mitragyna speciosa Korth.) side effects. J Ethnopharmacol. 2019;238:111876. doi: 10.1016/j.jep. 2019.11187649.
- Vicknasingam B, Chooi WT, Rahim AA, et al. Kratom and pain tolerance: a randomized, placebo-controlled, double-blind study. Yale J Biol Med. 2020;93:229-238.
- Fleming JH, Babyak CM, Alves EA. Analysis of heavy metals content in commercially available kratom products in Richmond, VA. Forensic Chem. 2023;33:100474.
- Long J. Kratom groups, researchers sound alarm over 7-hydroxymitragynine products. Natural Products Insider. June 12, 2024. Accessed January 5, 2025. https://www.naturalproductsinsider.com/herbs-botanicals/kratom-groups-researchers-sound-alarm-over-7-hydroxymitragine-products
- NDEWS. Weekly Monitoring Post. Issue 187. June 21, 2024. https://ndews.org/newsletter/weekly-briefing-issue-187/
- Todd DA, Kellogg JJ, Wallace ED, et al. Chemical composition and biological effects of kratom (Mitragyna speciosa): In vitro studies with implications for efficacy and drug interactions. Sci Rep. 2020:10:19158. https://doi.org/10.1038/s41598-020-76119-w
- 7-OHMZ Website. Certificate of Analysis. Accessed January 5, 2025. https://7ohmz.com/labs/
- Hydroxy Website. Certificate of Analysis. Accessed January 5, 2025. https://hydroxie.com/pages/lab-results
- Press’D Website. Certificate of Analysis. Accessed January 5, 2025. https://itspressd.com/wp-content/uploads/2024/04/CA240409-028-133-Alternative-Ventures-LLC-Pressd-1G-7-hydroxymitragynine-Tablets.pdf
- Gill LL. How to Shop for CBD. Consumer Reports. September 27, 2018. Accessed athttps://www.consumerreports.org/marijuana/how-to-shopfor-cbd/, April 13, 2019.
- Kamble SH, Obeng S, Leon F, et al. Pharmacokinetic and pharmacodynamic consequences of cytochrome P450 3A inhibition on mitragynine metabolism in rats. J Pharmacol Exp Ther. 2023;385:180-192. DOI: https://doi.org/10.1124/jpet.122.001525
- Kamble SH, León F, King TI, et al. Metabolism of a kratom alkaloid metabolite in human plasma increases its opioid potency and efficacy. ACS Pharmacol Transl Sci. 2020;3:1063-1068. doi: 10.1021/acsptsci.0c00075.
- Kratom.Clinicaltrials.gov. Accessed January 10, 2025.https://clinicaltrials.gov/search?term=kratom&limit=25&page=1
- 7-hydroxymitragynine.Clinicaltrials.gov. Accessed January 10, 2025.https://clinicaltrials.gov/search?term=7-hydroxymitragynine
- Matsumoto K, Horie S, Takayama H, et al. Antinociception, tolerance and withdrawal symptoms induced by 7-hydroxymitragynine, an alkaloid from the Thai medicinal herb Mitragyna speciosa. Life Sciences. 2005;78:2-7. https://doi.org/10.1016/j.lfs.2004.10.086.
- Hemby SE, McIntosh S, Leon F, Cutler SJ, McCurdy CR. Abuse liability and therapeutic potential of the Mitragyna speciosa (kratom) alkaloids mitragynine and 7-hydroxymitragynine. Addict Biol. 2019;24:874-885. doi: 10.1111/adb.12639.
- Wilson LL, Chakraborty S, Eans SO, et al. Kratom alkaloids, natural and semi-synthetic, show less physical dependence and ameliorate opioid withdrawal. Cell Mol Neurobiol. 2021;41:1131-1143. doi: 10.1007/s10571-020-01034-7.
- Smith KE, Boyer EW, Grundman O, McCurdy CR, Sharma A. The rise of novel, semi-synthetic 7-hydroxymitragnine products. Addiction. 2025;2:387-8. https://doi.org/10.1111/add.16728
- Reddit posts on 7-Hydroxymitragynine. Accessed January 5, 2025. 7_hydroxymitragynine (reddit.com)
- Edinoff AN, Sall S, Beckman SP, et al. Tianeptine, an Antidepressant with Opioid Agonist Effects: Pharmacology and Abuse Potential, a Narrative Review. Pain Ther. 2023;12(5):1121-1134. doi:10.1007/s40122-023-00539-5
- Samuels BA, Nautiyal KM, Kruegel AC, et al. The Behavioral Effects of the Antidepressant Tianeptine Require the Mu-Opioid Receptor. Neuropsychopharmacology. 2017;42(10):2052-2063. doi:10.1038/npp.2017.60
- Nelson S.Dozens of congressmen ask DEA not to ban kratom next week. US News & World Report, Sept 26, 2016.http://www.usnews.com/news/articles/2016-09-23/45-congressmen-ask-dea-not-to-ban-kratom-next-week (accessed 2016 Nov 17).
36. Kratom Regulation Map. Global Kratom Coalition. Accessed January 15, 2025. https://globalkratomcoalition.org/regulation-map
- Is Kratom Legal? A state-by-state guide. Accessed January 5, 2025. https://www.sprouthealthgroup.com/substances/is-kratom-legal-by-state/
38. Kratom Consumer Advisory Council. Global Kratom Coalition. Accessed January 15, 2025. https://globalkratomcoalition.org/kcac
- Press’d Website. The Hulk Blend Product. Accessed January 5, 2025. https://itspressd.com/product/hulk-blend-30mg/
40. Mitragynine Pseudoindoxyl 5ct Chewable Tablets! Legal Herb Shop. Accessed January 15, 2025. https://legalherbalshop.com/product/mitragynine-pseudoindoxyl-5ct-chewable-tablets/
41. Eat Ohmz 7-OH + 8-OH + Red-OH Extract Tablets Full Case 30mg. Great CBD Shop. Accessed January 15, 2025. https://greatcbdshop.com/product/eat-ohmz-7-oh-8-oh-red-oh-extract-tablets-full-case-30mg/
- White CM. ‘Gas Station Heroin’ Is a Dangerous and Often Contaminated Supplement. Scientific American. February 6, 2024. Accessed January 5, 2025. https://www.scientificamerican.com/article/gas-station-heroin-is-a-dangerous-and-often-contaminated-supplement/
- KCAC Position Statement: Kratom Use in Children. Kratom Consumer Advisory Council. November 4, 2024. Accessed January 15, 2025.https://static1.squarespace.com/static/6508b3f79033221c2aa1ea17/t/677c4aed329dad0ce52dfd8a/1736198893809/KCAC+Positioning+Doc_1+Final.pdf
- California Legislature. Kratom Consumer Protection Act Bill Number CA AB2365. Accessed January 5, 2025. https://legiscan.com/CA/text/AB2365/id/2962565
- López G, Orchowski LM, Reddy MK, et al. A review of research-supported group treatments for drug use disorders. Subst Abuse Treat Prev Policy. 2021;16(1):51. doi: 10.1186/s13011-021-00371-0.