Learning Objectives
After completing this application-based continuing education activity, pharmacists will be able to
| · Outline the causes of pet allergies in dogs, cats, and other less common species |
| · Differentiate between allergic sensitization, allergy, and cross sensitivity |
| · Paraphrase facts that prove that currently, hypoallergenic dogs and cats are a myth |
| · Compare nonpharmacologic, over the counter, and prescription treatments in terms of dosing, effectiveness, and cost |
After completing this application-based continuing education activity, pharmacy technicians will be able to
| · Outline the causes of pet allergies in dogs, cats, and other less common species |
| · Differentiate between allergic sensitization, allergy, and cross sensitivity |
| · Paraphrase facts that prove that currently, hypoallergenic dogs and cats are a myth |
| · Identify patients whose complaints indicate they may need referral to a pharmacist |

Release Date: March 25, 2026
Expiration Date: March 25, 2029
Course Fee
FREE
There is no grant funding for this CE activity
ACPE UANs
Pharmacist: 0009-0000-26-020-H01-P
Pharmacy Technician: 0009-0000-26-020-H01-T
Session Codes
Pharmacist: 23YC08-JKT44
Pharmacist Technician: 23YC08-TKX48
Accreditation Hours
2.0 hours of CE
Accreditation Statements
| The University of Connecticut School of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education. Statements of credit for the online activity ACPE UAN 0009-0000-26-020-H01-P/T will be awarded when the post test and evaluation have been completed and passed with a 70% or better. Your CE credits will be uploaded to your CPE monitor profile within 2 weeks of completion of the program. |  |
Disclosure of Discussions of Off-label and Investigational Drug Use
The material presented here does not necessarily reflect the views of The University of Connecticut School of Pharmacy or its co-sponsor affiliates. These materials may discuss uses and dosages for therapeutic products, processes, procedures and inferred diagnoses that have not been approved by the United States Food and Drug Administration. A qualified health care professional should be consulted before using any therapeutic product discussed. All readers and continuing education participants should verify all information and data before treating patients or employing any therapies described in this continuing education activity.
Faculty
Yangzhou (Marina) Li, MS, PharmD
Medical Writing Scientist
Janssen of Pharmaceutical Companies of Johnson and Johnson,
Boston, MA
Dylan DeCandia, PharmD
Freelance Medical Writer
Franklyn's Pharmacy
Ho-Ho-Kus, NJ
Faculty Disclosure
In accordance with the Accreditation Council for Pharmacy Education (ACPE) Criteria for Quality and Interpretive Guidelines, The University of Connecticut School of Pharmacy requires that faculty disclose any relationship that the faculty may have with commercial entities whose products or services may be mentioned in the activity.
Dr. Li is a full time employee of Janssen Pharmaceutical of Johnson and Johnson and previously worked for Nest Bio and LegendBiotech. Dylan DeCandia does not have any relationships with ineligible companies and therefore has nothing to disclose.
ABSTRACT
Many American households have pets, and many others would like to have pets but family members have pet allergies. Allergies to cats and dogs are common (an estimated 15% to 30% of people are allergic to companion pets), and allergies to unusual or exotic pets have increased over the last decade. Pet allergy is an allergic reaction to proteins (allergens) found in animals’ skin cells (dander), saliva, urine, or sweat on their fur. Most animal allergens belong to one of three primary protein families. Pet allergies are currently incurable. The treatment goal is to control symptoms and improve patients’ functional status and well-being. Options include nonpharmacologic interventions like cleaning and bathing the pet and pharmacologic management with antihistamines, corticosteroids, anticholinergic nasal sprays, mast cell stabilizers, or leukotriene modifiers. Allergists will consider allergy-specific immunotherapy when medications and/or avoidance measures fail.
CONTENT
Content
INTRODUCTION AND EPIDEMIOLOGY
The American Pet Products Association (APPA) estimates that approximately 70% of Americans keep pets in their household, equating to 90.5 million homes. Dogs and cats are the most popular and live in around 69.0 and 45.3 million United States (U.S.) households, respectively, followed by 11.8 million households for freshwater fish, 9.9 million households for birds, and 3.5 million households for horses.1 Public, residential, leisure, and specific occupational environments (e.g., farms, laboratories, pet shops) have high concentrations of pet allergens because of the high prevalence of community pet-keeping and Americans’ tendency to live indoors. Allergic reactions to pets have been recognized for at least 100 years.2 Risk factors for developing asthma and rhinitis include allergies to furry animals, especially cats and dogs.3 Direct or second-hand pet exposure increases the likelihood of exacerbating disease in pet-sensitive people. However, evidence also shows that early childhood exposure to dogs or cats before one year of age may have protective effects in preventing allergic sensitization.4
Notably, allergies to unusual or exotic pets have increased over the last decade.5 In many urban areas, apartment complexes prevent owning large pets or charge a fee for owning cats and dogs, leading to the choice of smaller, more unusual animals. Some examples of uncommon pets are rodents (mice, rats [which allegedly make very good pets], guinea pigs, and other mammals like ferrets, pigs), amphibians (axolotl [a Mexican salamander], dart frogs, and fire belly newts), and reptiles (snakes).6 The allergic signs and symptoms or diseases associated with uncommon pets are like those manifested in cat and dog allergies. In addition, patients may present with respiratory symptoms induced by bird allergens and gastrointestinal symptoms after consuming bird eggs; this is called a bird-egg syndrome.7
Overall, the incidence of specific allergy to exotic or uncommon pets is unknown because literature only includes isolated cases or small series. In the U.S., an estimated 15% to 30% of people are allergic to their pets.8 Among people with pet allergies, a fraction is sensitized to more than one animal. Moreover, according to the Asthma and Allergy Foundation of America, cat allergies are reported twice as often as dog allergies. Animals are also recognized as the third leading cause of allergic asthma, after mites and pollens.8 Many people adopt ferrets or rabbits, believing they are hypoallergenic. They are not, and pharmacy staff should be aware of that fact.9,10 The most frequent allergic reactions result from inhalation, contact, or bites.
This continuing education activity summarizes knowledge of pet allergens, including those from uncommon pets; the allergy reaction mechanism and its signs and symptoms; current advances in diagnosis and treatment methods such as immunotherapy; and recommendations for patient education and counseling.
PAUSE AND PONDER: When patients ask about medication for pet allergies, what kinds of questions should you ask?
PET ALLERGENS
Allergy Mechanisms
Compared with other conditions’ mechanisms, allergy mechanisms are simple and encompass three specific paths: allergic sensitization, allergy, and cross-reactivity.11
- Allergic sensitization is the presence of immunoglobulin E (IgE) antibodies to an allergen.
- Allergy is the occurrence of reproducible symptoms or signs initiated by exposure to a defined stimulus at a dose tolerated by nonallergic persons and mediated by specific immunologic mechanisms. If no symptoms develop, a person could be sensitizing to a particular allergen but not be allergic.
- Cross-reactivity is the process of IgE antibodies (originally developed against a given allergen) binding to homologous molecules originating from a different allergen source.
Characterizing Pet Allergens
Allergies to pets are common. Pet allergy is an allergic reaction to proteins (allergens) found in animals’ skin cells (dander), saliva, urine, or sweat on their fur.5 Allergens within the same protein family can cause cross-reactivity. Most allergens are spread via airborne particles. Dander contains allergens formed in sebaceous gland secretions and saliva. Secretions containing allergens adhere to the hair and stratum corneum of the skin. When an animal sheds, tiny particles disperse into the air and remain buoyant for an extended period of time. After the particles slowly settle onto the floor, furniture, or other items, they can be easily re-dispersed into the air. As a result, pet-sensitive people could experience allergy symptoms in the nose, eyes, and respiratory tract even if the pet is not present.5 Additionally, people can carry pet allergens that settled onto their clothing or hair.
For cats and dogs, the primary allergen sources are dander and saliva. Similarly, the primary allergen source in rabbits is saliva. In contrast, the primary allergen source is urine in rodents (mice and rats) and Mustelidae (ferrets and minks).
Rodents are an interesting case study. Most research laboratories experience a very high rate of staff turnover because lab workers develop allergies to rodents. Children who are exposed to rodent urine can develop this allergy, too. Male rodents produce a larger quantity of and more condensed urine than female rodents. This explains why people who commonly come in contact with male rodents are more likely to develop allergic symptoms. Allergy to rodents acts as an occupational hazard for researchers. Mouse urine is the most concentrated of all urines—far more concentrated than any other species.12 One study showed that 30% of people exposed to mice and 13.7% of people exposed to rats suffered from allergy symptoms.12 Symptoms range from conjunctivitis to asthma to skin reactions, which makes working with these animals difficult.
Most animal allergens belong to one of three primary protein families. Within the three families, lipocalin-like proteins and the serum albumin family are the two most widely studied. Other identified allergens are considered minor, including gelatins, immunoglobulins, and transferrins presented in secretions and dandruff. Knowledge of these allergens’ allergenicity and cross-activity is essential to improve treatment and prevent allergic reactions. Table 1 summarizes partially characterized pet allergens, including those generated by exotic pets, because not all allergens are fully characterized.5
Table 1. Summary of Characterized Pet Allergens13-22
| Common Name of Animal | Source | Allergen | Family |
| Dog | Dander, saliva, hair | Can f 1 (major allergen)
Can f 2 Can f 4 Can f 6 Can f 3 Can f 5 Can f 7 Can f 8 |
Lipocalin
Lipocalin Lipocalin Lipocalin Albumin Arginine esterase (kallikrein) Epididymal secretory protein E1 or Niemann Pick type C2 protein Cystatin |
| Cat | Sebaceous, anal, and salivary gland | Fel d 1 (major allergen)
Fel d 2 Fel d 4 Fel d 7 Fel d 3 Fel d 5w Fel d 8 Fed d 6w |
Uteroglobin
Albumin Lipocalin Von Ebner gland protein Cystatin Cat IgA Latherin-like IgM |
| Horse | Dander, sublingual, submaxillary salivary glands, and urine | Equ c 1 (major allergen)
Equ c 2 Equ c 4 Equ c 3 Equ c 6 |
Lipocalin
Lipocalin Latherin Albumin Lysozyme |
| Chinchilla | Epithelia, saliva, urine | Chi La
Chi Lb |
Protein kinase inhibitor
Lipocalin |
| Guinea pig
|
Cav p 1 (major allergen)
Cap p 2 (major allergen) Cap p 3 Cap p 4 Cap p 6 |
Lipocalin
Lipocalin Lipocalin Serum albumin Lipocalin |
|
| Gerbil | Epithelial, salvia, urine, sleep bed | Mer un 23kDa
Mer un 4 |
Lipocalin
Serum albumin |
| Siberian hamster | Epithelial, saliva, urine | Phod s 1 | Lipocalin |
| Rat | Rat n 1 (major allergen)
Rat n 4 Rat n 7 |
Lipocalin; alpha-2u-glubulin
Serum albumin Immunoglobulin |
|
| Mouse | Mus m 1 (major allergen)
Mus m 2 Mus m 4 Mus m 7 |
Lipocalin; urinary prealbumin
Unknown Serum albumin Immunoglobulin |
|
| Rabbit | Ory c 1
Ory c 2 Ory c 3 Ory c 4 |
Lipocalin
Lipocalin Secretoglobin Lipocalin |
|
| Ferret | Mus p 17
Mus p 66 |
Unknown
Serum albumin |
|
| Pig | Meat | Sus s 1
Sus s 5 Sus s 6 |
Serum albumin
Lipocalin Serum albumin |
Lipocalin Superfamily
More than 50% of allergens identified from furry animals belong to the lipocalin superfamily and are found in animal dander, saliva, and urine.23 Lipocalins are large proteins and can induce IgE production in a large proportion of atopic individuals (people who have enhanced immune response to common allergens) who are exposed to the allergen source.24
Serum Albumin Family
Serum albumin is a globular protein prone to participation in IgE-mediated cross-reactions.24 Serum albumin is commonly found in pet dander and saliva and causes an allergic reaction by inhalation and ingestion.
Secretoglobin Superfamily
Secretoglobins are the most potent allergens in cats (e.g., Fel d 1) and other pets (e.g., rabbit; Ory c 3). Produced by the skin, salivary and lacrimal glands, these proteins have an unknown function. Dried saliva and dandruff are spread as airborne particles and cause sensitization in susceptible people.25
SIGNS AND SYMPTOMS OF PET ALLERGIES
The most frequently observed pet allergies result from inhalation, contact, and bites. The main allergic symptoms are similar across both common and uncommon pet types. They present as rhinitis, conjunctivitis, urticaria (red, itchy welts that result from a skin reaction), and lower and upper respiratory symptoms, which can be mild to severe and rarely cause anaphylactic shock.5
Hypoallergenic Pets
“Hypoallergenic” is defined as possessing decreased risk of causing an allergy in people, which means that hypoallergenic animals could still elicit allergies in humans.9 To make hypoallergenic animals, breeders or researchers combine breeds that produce less allergen (in dogs, breeders use breeds that shed less than other breeds, or have hair rather than fur). However, animals often have different mechanisms of allergenicity, so infrequent shedding does not solve all allergy problems.
In a dog allergen study, homes that included hypoallergenic dogs had no statistically significant difference in dog allergen levels compared to homes that included non-hypoallergenic dogs. The common allergen in dogs, Can f 1, was reported at similar levels in all groups.25 The frequency of shedding varies in different dog breeds, but all dogs can elicit allergies in humans.
The main allergen in cats, Fel d 1 protein, comes from their saliva and sweat glands. Because of its small size and adhesiveness, Fel d 1 floats around and sticks to everything, making it almost impossible to remove physically. In fact, Fel d 1 measures in at less than one-tenth the size of ribosome; it’s so small, it easily navigates its way deep into the lungs and can precipitate asthma.26 For this reason, making a completely hypoallergenic cat has proven impossible, however vaccines to decrease the production of Fel d 1 protein have been studied; one vaccine is a combination of recombinant Fel d 1, tetanus toxoid protein, and a snippet of the coat of a plant virus.27 Researchers are unsure as to the purpose of Fel d 1 in cats or why levels of Fel d 1 vary.
Ferrets—which are related to otters, minks, and weasels—are considered hypoallergenic because they are less likely to cause an allergic reaction compared to other animals. However, they can still provoke allergies in people. Allergies to ferrets come from their hair, saliva, and urine. Ferret hair and saliva is usually easy to control because they shed infrequently and do not lick people like dogs and cats often do. However, urine is harder to control and can cause allergies when owners clean crates.9
Rabbits produce allergens through dander, hair from shedding, and saliva. They tend to shed more often than ferrets, around every three months, so keeping up with cleaning may be difficult. Rabbit hair isn’t naturally allergenic, but when rabbits lick their fur, they transfer a saliva protein that is contaminated with the protein allergen.10
DIAGNOSIS
Skin Prick Test
Allergists (allergy specialists) use skin prick tests together with medical history and physical examinations to rule out or confirm a suspected IgE-mediated animal allergy.28 Manufacturers prepare skin prick tests by extracting natural allergens from animal hair, dander, and urine. The doctor or nurse will prick the patient’s skin on the forearm or upper back and determine if an allergic reaction occurs within 15 minutes. If a patient develops a red, itchy bump where the pet allergen extract is pricked into the skin, the patient is allergic to that pet allergen. Diagnosticians should first use a skin prick test as it is inexpensive, easy to use, and quick to perform. However, allergen concentrations and components are inconsistent, varying among similar commercial tests from different manufacturers. Healthcare providers should be aware that patients’ test results may be inconsistent if they use different skin prick tests at different times.28
Serum-specific IgE Test
Allergists can use a serum-specific IgE (blood) test when patients’ symptoms and skin test results are contradictory or when patients’ skin conditions prevent a skin test. Serum-specific IgE tests can only determine if a patient is sensitized to a specific pet allergen, but it cannot determine if a patient is allergic to that allergen. Serum-specific IgE tests are highly sensitive, but prone to false-positive results. From this perspective, serum-specific IgE tests may be less accurate than skin prick tests.29
Molecular Diagnosis
Recent scientific advances have allowed molecular diagnosis to differentiate patients who are allergic to a single species or sensitized due to cross-reactivity. This method can aid targeted recommendations for avoidance and assess the choice and composition of immunotherapy.28
PET ALLERGY MANAGEMENT
Pet allergies cannot currently be cured. The treatment goal is to control symptoms and improve patients’ functional status and well-being.
Nonpharmacologic Treatment – Avoid & Minimize Allergen Exposure
Current recommendations for managing pet allergy symptoms start with exposure avoidance. Starting when animals are young, bathing them at least once weekly can reduce allergens and eliminate reactions in humans who are exposed to them (see SIDEBAR).30 Immediate removal of animals from the household will not alleviate symptoms if the owner has carpeting and other pieces of furniture/items that the pet slept or sat on. Mammalian allergens are stable and can persist in house dust for up to six months.32 Additionally, using high-efficiency particulate air (HEPA) filters and mattress encasement, vacuuming, and chemically treating carpet are alternative methods for reducing exposure to contaminated materials, but may not reduce disease severity.33
PAUSE AND PONDER: When patients have pet allergies, which symptoms are best treated with antihistamines?
SIDEBAR: To Bathe or Not to Bathe…26,31
Bathing a cat or dog regularly appears to reduce the quantity of allergen harbored by the pet. To effectively lower Can f 1 concentrations, owners need to bathe the animal at least twice every week because Can f 1 concentrations rise rapidly, approaching baseline concentrations within three days after washing. Twice-weekly bathing can reduce the amount of recoverable Can f 1 on dogs by more than 80%, but researchers note that ideally, one would bathe the dog two to three times every week. Airborne Can f levels can fall by ruff-ly 40% but will quickly escalate.
However, the beneficial effects of reducing allergen levels by regular bathing are more likely associated with dogs, because their allergen burden returns faster than that of cats. So, bathing animals reduces the amount of allergen far better than vacuuming.
But should companion animals be bathed so often?
Most cats are notoriously averse to bathing, although some breeds like water (i.e., the Bengal). Dogs vary in the response to bathing—some like it, others do not. People who plan to bathe their cats or dogs regularly should do three things:
- Check with a veterinarian or a breed advocacy group. The American Kennel Club indicates that how often an owner should bathe a dog depends on the dog’s coat type and presence or absence of an undercoat (in the latter case, frequent bathing can affect a dog’s temperature regulation). Bathing an animal is not just about a human’s allergies, the animal’s health and welfare should be a primary concern.
- Consider the labor and time involved in bathing a pet often, safely, and well.
- Start when the animal is young.
An allergen reducing cat food (Pro Plan LiveClear) is now available, and its manufacturer indicates it reduces the number of allergens in cat hair and dander by 47% after three weeks of feeding.34 It is produced using eggs that contain an anti-Fel d1 antibody. When cats consume the food, the egg powder binds to and neutralizes Fel d1 in the cat’s saliva.34
Pharmacologic Treatment
When avoidance and reducing allergens are not enough, depending on the severity of signs, over the counter (OTC) medications like antihistamines or local/topical steroids may provide temporary relief of allergy symptoms.35 Those symptoms include runny/itchy nose or throat, sneezing, and itchy, red or watery eyes. Combination products that contain both an antihistamine and a decongestant or an analgesic are available but should be used with caution due to the increased risk of adverse effects. Other allergy medications, besides the ones mentioned, are used less often, including mast cell stabilizers and leukotriene antagonists. Table 2 summarizes common medications (both OTC and prescription) for treating mild to moderate allergy symptoms.35
Table 2. Medications to Treat Allergy Symptoms36
| Medication | Mechanism of Action | Adverse Effects | Notes |
| Antihistamines | |||
| 1st generation (nonselective, more sedating):*
diphenhydramine, chlorpheniramine, clemastine 2nd generation (less sedating, less drowsiness): cetirizine,* desloratadine,* fexofenadine,* levocetirizine,* and loratadine* Azelastine has nasal spray* and eye drop formulations. Epinastine and olopatadine* are formulated as eye drops. |
Blocks histamine and its binding to receptors, prevents histamine-caused redness, swelling, itching, and changes in secretions during an allergic response | · Drowsiness
· Fatigue · Headache |
The 2nd generation antihistamines are preferred over 1st generation based on safety and efficacy data.
|
| Corticosteroids | |||
| Available as tablets, liquids, nasal sprays, topical creams for skin allergies, topical eye drops for conjunctivitis.
Some steroids include: beclomethasone, ciclesonide, fluticasone furoate,* mometasone, budesonide,* triamcinolone,* dexamethasone ophthalmic, prednisone, etc. |
Anti-inflammatory effect | Short-term use:
Weight gain, fluid retention, high blood pressure
Long-term use: Growth suppression, diabetes, cataracts of the eye, osteoporosis, muscle weakness
Side effects of inhaled steroids: Cough, hoarseness, fungal infection of the mouth
|
Highly effective for allergies but must be taken regularly. It may take 1 to 2 weeks before the full effect. |
| Decongestants | |||
| Available as nasal sprays, eye drops, liquids, and tablets
Some decongestants include: pseudoephedrine,* phenylephrine,* and oxymetazoline* nasal sprays
|
Shrinks swollen nasal tissues and blood vessels to relieve the symptoms of nasal swelling, congestion, mucus secretion, and redness | · Increased blood pressure
· Insomnia · Anxiety, feeling nervous, restlessness |
Relieve congestion and are often prescribed with antihistamines for allergies.
Contraindicated in patients with severe coronary artery disease, severe hypertension, and who concomitantly use monoamine oxidase inhibitors
Short-term use only (~5 days). Long-term use can make symptoms worse. |
| Combination Allergy Drugs | |||
| Some combination drugs include:
cetirizine/pseudoephedrine,* fexofenadine/ pseudoephedrine,* diphenhydramine/ pseudoephedrine,* loratadine/pseudoephedrine,* pseudoephedrine/triprolidine* for nasal allergies, and naphazoline/pheniramine* for allergic conjunctivitis
|
Effects from each component | Side effects from each component | Use with caution due to increased risk of adverse effects |
| Anticholinergic Nasal Spray | |||
| Ipratropium bromide nasal spray to control nasal discharge | Antisecretory properties in the nasal mucosa | · Bitterness of the mouth
· Dry nose, nosebleeds, or irritation · Dizziness · Headache · Sore throat · Respiratory tract infection |
Some patients may feel better right away. For others, it may take 1 to 2 weeks before the medicine helps. It is important for patients to continue use of this medication as instructed. |
| Mast Cell Stabilizers | |||
| Available as eye drops for allergic conjunctivitis and nasal sprays for nasal allergy symptoms
Some mast cell stabilizers include cromolyn sodium,* iodoxamide-tromethamine, nedocromil, pemirolast, etc. |
Prevents histamine release from mast cells | · Throat irritation, coughing, skin rashes
· Eye drops may cause blurred vision, stinging, and burning |
For mild to moderate symptoms
Not as effective as steroids |
| Leukotriene Modifiers | |||
| Montelukast:
Indicated for adults and pediatric patients six months or older with perennial allergic rhinitis. May be less effective than loratadine or cetirizine for reducing daytime nasal symptoms |
Montelukast binds to leukotriene receptors in the human airway (smooth muscle cells and macrophages), preventing airway edema, smooth muscle contraction, and other respiratory inflammation | · Stomach pain or upset
· Headache · Stuffy nose · Cough · Fever · Rash · Irritability |
Warn patients to report behavior changes, including suicidal ideation or suicidal behavior
Avoid concomitant use of aspirin or NSAIDs in aspirin-sensitive patients |
| *Indicates over-the-counter (OTC) medication | |||
In general, for conditions eligible for self-care (e.g., allergic rhinitis) patients should start taking OTC allergy medications one week before they expect symptoms from a predictable exposure or as soon as possible before allergen exposure (for episodic exposure).35 Prescribers should tailor the pharmacologic therapy and length of treatment based on symptoms and severity. Usually, complete relief takes two to four weeks. Intranasal steroids control nasal symptoms more effectively than antihistamines, as they inhibit multiple cell types and mediators, and should be recommended for moderate or persistent allergic rhinitis. Decongestants are effective in nasal congestion but have little effect on other symptoms. Intranasal and ocular preparations are available for nasal and eye symptoms. Intranasal cromolyn is the preferred initial choice for pregnant or lactating patients, as the body does not absorb it based on the route of administration. As mentioned in the table, fluticasone and triamcinolone nasal sprays are available OTC.35
If a patient has persistent allergies, allergy medication is more effective when taken regularly.35 For example, if a patient with moderate or severe persistent allergic rhinitis has completed two to four weeks of treatment with intranasal corticosteroids or oral antihistamines and achieved symptomatic control, healthcare providers can optimize the treatment’s effect by reducing the dose and continuing treatment for one additional month. If a patient’s symptoms are uncontrolled after two to four weeks of OTC treatment, pharmacists should assess the patient’s adherence and refer for prescription therapy if necessary.35
PAUSE AND PONDER: Which providers in your area provide allergen-specific immunotherapy? What should patients expect if they take this route?
Allergy Immunotherapy
Allergen-specific immunotherapy has been used in pet allergies for years and has proven efficacy to help control symptoms and prevent disease progression. Allergists will consider allergy-specific immunotherapy when symptoms are uncontrolled by medications and/or avoidance measures, when adverse drug effects are intolerable, or when patients want to reduce long-term use of allergy medications.37
The basis for allergen-specific immunotherapy is gradual reprogramming of the immune system to build a tolerance to allergens. The U.S. Food and Drug Administration (FDA) characterizes allergen-specific immunotherapies as biologics because they are produced from living cells, not synthesized by chemists, and regulated under the Center for Biologics Evaluation and Research (CBER).38 This class comes in three forms:
- Sublingual allergy immunotherapy (SLIT) tablets
- SLIT drops, and
- subcutaneous allergy immunotherapy (SCIT)
As of 2022, the FDA has approved four SLIT tablets to treat allergic rhinitis with or without allergic conjunctivitis caused by ragweed, northern pasture grasses, and dust mites in susceptible individuals; the FDA has not approved SLIT tablets for pet allergies.22
SLIT drops are made from FDA-approved allergy extracts used to make SCIT shots. However, these extracts are only FDA-approved for injection use under the skin, and they are not approved for use under the tongue. Therefore, SLIT drops are not FDA-approved and are off-label in the U.S., and Medicare or Medicaid does not cover these treatments in most cases. Despite not having FDA approval, patients can still receive SLIT drops from some prescribers who prepare a custom-mixed formulation but must pay out of pocket. Research indicates SLIT is safe and effective.39
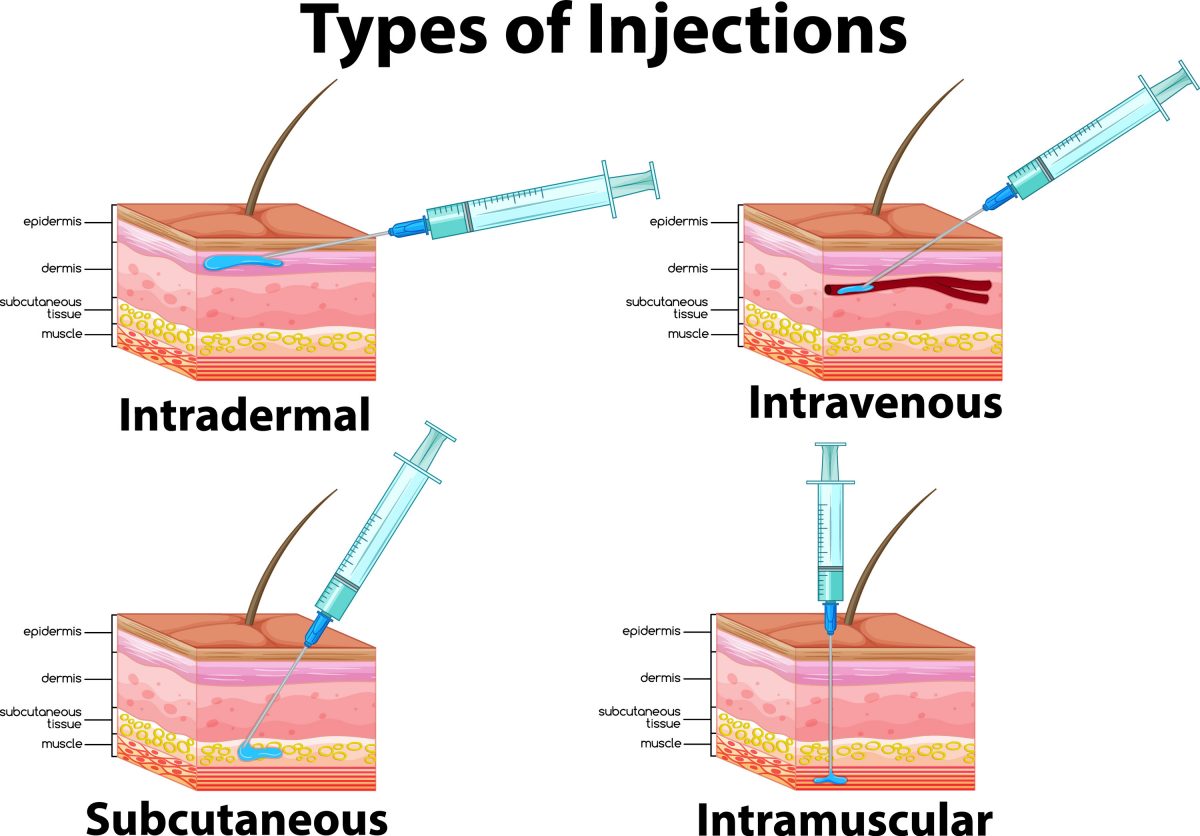
The FDA has approved SCIT for cat allergies, but not for other pet allergies. Patients who receive SCIT usually call it “allergy shots.” One systemic review evaluated 88 trials that enrolled 3,459 asthmatic patients and exposed them to SCIT. One case of deterioration in asthma symptoms was avoided for every three patients treated with SCIT (95% CI, 3-5), and one patient would avoid increasing symptomatic medication use for every four patients treated (95% CI, 3-6).40 Another study found that SCIT can reduce the need for systemic steroids in allergic rhinitis patients.41 Usually, the patient receives a solution for injection with 10,000 bioequivalent allergy units (BAUs) per milliliter (standardized extract) of lyophilized cat hair and dander added to glycerol and human serum albumin (0.03%). A clinician administers one to two subcutaneous injections every week starting at low doses (1:10,000 dilution) and titrating up to a seemingly effective maintenance dose. Then, the prescriber extends the injection interval gradually to every 2 weeks to 4 weeks. For cat allergens, the effective maintenance dose usually falls within the 1,000 to 4,000 BAU range.42
S'CIT sometimes can cause treatment-related systemic allergic reactions; however, near-fatal or severe reactions are rare, and most reactions are local and mild (swelling, pruritis, and redness at injection site).43 SCIT should not be recommended to patients who have severe uncontrolled heart problems or asthma if they take beta-blockers, which are associated with more frequent reactions, more severe reactions, and reactions that are refractory to epinephrine. Additionally, allergy shots should not be recommended for pregnant women unless discussed with their obstetricians.43
Both SCIT and SLIT require gradual up-titration of dosages with ongoing and multiple treatments and may take three to five years to reach desensitization. Also, for SCIT, based on its route of administration (subcutaneous injections are invasive), patients will need to visit the doctor's office more frequently and may experience the treatment-associated side effects.
SLIT has been increasingly recommended because of its ability to modify the immune system for the long term while reducing allergy symptoms. SLIT also showed a safer profile, only associated with mild mouth symptoms, and improved adherence compared to SCIT.44 When compared to traditional allergy treatments, SLIT tablets showed similar clinical efficacy to nasal corticosteroids and greater clinical efficacy than second-generation antihistamines and montelukast.45
What About Cost?
In adherent patients, SCIT and SLIT have proven to be an economically viable option. The annual cost of using SCIT depends on patients’ insurance: Medicare ($1021.70), Medicaid ($758.16), and the commercial average ($1722.24). Yearly treatment costs for SLIT are self-pay because treatment is not FDA approved and costs around $679.25.46 Because SLIT drops are administered at home by patients, they tend to be more affordable than the cost of SCIT. Patient preference might be for a once monthly administration, rather than taking oral antihistamines daily.
OTC medications are less expensive than immunotherapy, but costs vary. In a comparison of second-generation antihistamines versus montelukast, levocetirizine (Xyzal) had the best efficacy per cost value. Generic fexofenadine (Allegra), although similar in efficacy, was more expensive than levocetirizine.44
CONCLUSION
Healthcare providers should counsel patients about reducing allergen exposure and help patients to choose OTC medications for self-care based on individual patient needs and conditions to optimize treatment effects. Pharmacy staff should refer patients to allergists when necessary to identify the cause of their allergy symptoms. If a patient's allergy does not allow him or her to have pets at home and the patient owns a pet, suggest that the patient ask family members or friends about placement before contacting the local animal shelters.
Pharmacist Post Test (for viewing only)
Pet Allergies
Pharmacist Post-test
After completing this continuing education activity, pharmacists will be able to
1. Outline the causes of pet allergies in dogs, cats, and other less common species
2. Differentiate between allergic sensitization, allergy, and cross sensitivity
3. Paraphrase facts that prove that currently, hypoallergenic dogs and cats are a myth
4. Compare nonpharmacologic, over the counter, and prescription treatments in terms of dosing, effectiveness, and cost
1. What is the major allergen in dogs?
A. Can f 3
B. Can f 1
C. Fel d 1
2. When comparing allergy immunotherapies (SCIT, SLIT) and traditional allergy treatments, how do they differ?
A. Traditional allergy treatments are more clinically efficacious, SCIT and SLIT therapies should not be considered in treatment
B. Traditional allergy treatments are more effective, but with the recent FDA approval of SLIT, it should be considered more often
C. Although not FDA approved, SLIT showed similar clinical efficacy to nasal corticosteroids and more clinical efficacy to second-generation antihistamines in trials
3. What is the best way to define hypoallergenic animals?
A. Animals that are less likely to cause allergies in humans.
B. Animals that cannot cause allergies in humans
C. Animals that do not cause conjunctivitis, but other common symptoms may still occur
4. A mother brings her young son to the pharmacy and says that the allergist indicates he has an allergy to their cat. She asks what this means. What is the BEST answer?
A. Her son has immunoglobulin G (IgG) antibodies to an allergen.
B. Her son will experience reproducible symptoms when exposed to the cat.
C. Her son will have symptoms when exposed to any furry animal.
5. Lance, a college student who lives in a group house, comes in and says that he has tried several medications for allergic symptoms linked to his roommate’s three cats. The medications relieved the symptoms but caused so much drowsiness, he couldn’t study. His allergist is now recommending he start immunotherapy. What is the MOST LIKELY reason the allergist is making this recommendation?
A. Lances’ symptoms are uncontrolled by medications
B. Lance is experiencing intolerable adverse effects
C. Lance want to reduce his use of allergy medications.
6. Lance returns to the pharmacy to pick up his atenolol for hypertension and he said the allergist has asked him to decide if he wants to take SCIT or SLIT. He asks you which factors he should consider. What is the BEST answer?
A. Advise him to consider cost, dosing frequency, and route of administration
B. Advise him to consider cost and convenience alone as they are both effective
C. Advise him to tell his allergist he is taking a beta blocker, so SLIT is preferred
7. Emily and her mom come to the pharmacy and they are very excited. They are considering adopting a dog! Emily has asthma and multiple allergies, and the pediatrician has told them she is probably allergic to or will become allergic to dogs. As Mom chatters, she tells you that the 9-year-old dog, Raven, is an Alaskan Malamute (a breed that has a heavy undercoat) that weighs 95 pounds. She said that a friend told her that if she washes the dog two or three times a month, allergies will not be a problem. She says, “I think I can find time to wash a dog twice a month.” What is the MOST IMPORTANT FACT you should bring to her attention?
A. Before adopting Raven, check with a veterinarian or a breed advocacy group to determine if bathing is a good idea.
B. Bathing a pet two to three times a month is not frequent enough to reduce the allergen load—you have to bathe them two to three times a week.
C. Look for a younger Alaskan Malamute—maybe a puppy—so the dog will get used to being bathed so often.
8. Adele, who is 7 months pregnant, is experiencing an allergic reaction to a visiting ferret. She asks you to recommend an OTC product to reduce her nasal stuffiness and itchy eyes. Which is the BEST product to recommend?
A. Intranasal cromolyn
B. Oral levocetirizine
C. Oral diphenhydramine
9. Which of the following have similar effectiveness for pet allergies, but different cost effectiveness?
A. Fluticasone and fexofenadine
B. Montelukast and loratadine
C. Levocetirizine and fexofenadine
10. Which medication class should be used for no longer than five days at a time?
A. Decongestants
B. First generation antihistamines
C. Nasal corticosteroids
Pharmacy Technician Post Test (for viewing only)
Pet Allergies
Pharmacy Technician Post-test
After completing this continuing education activity, pharmacy technicians will be able to
• Outline the causes of pet allergies in dogs, cats, and other less common species
• Differentiate between allergic sensitization, allergy, and cross sensitivity
• Paraphrase facts that prove that currently, hypoallergenic dogs and cats are a myth
• Identify patients whose complaints indicate they may need referral to a pharmacist
1. What is the major allergen in dogs?
A. Can f 3
B. Can f 1
C. Fel d 1
2. When comparing allergy immunotherapies (SCIT, SLIT) and traditional allergy treatments, how do they differ?
A. Traditional allergy treatments are more clinically efficacious, SCIT and SLIT therapies should not be considered in treatment
B. Traditional allergy treatments are more effective, but with the recent FDA approval of SLIT, it should be considered more OFTEN?
C. Although not FDA approved for pet allergies, SLIT showed similar efficacy to nasal corticosteroids and more clinical efficacy to second-generation antihistamines in trials
3. What is the best way to define hypoallergenic animals?
A. Animals that are less likely to cause allergies in humans.
B. Animals that cannot cause allergies in humans
C. Animals that do not cause conjunctivitis, but other common symptoms may still occur
4. A mother brings her young son to the pharmacy and says that the allergist indicates he has an allergy to their cat. She asks what this means. What is the BEST answer?
A. Her son has immunoglobulin G (IgG) antibodies to an allergen.
B. Her son will experience reproducible symptoms when exposed to the cat.
C. Her son will have symptoms when exposed to any furry animal.
5. Lance, a college student who lives in a group house, comes in and says that he has tried several medications for allergic symptoms linked to his roommate’s three cats. The medications relieved the symptoms but caused so much drowsiness, he couldn’t study. His allergist is now recommending he start immunotherapy. What is the MOST LIKELY reason the allergist is making this recommendation?
A. Lances’ symptoms are uncontrolled by medications
B. Lance is experiencing intolerable adverse effects
C. Lance want to reduce his use of allergy medications.
Links to LO #4 Answer found on page 10
6. Lance returns to the pharmacy to pick up his atenolol for hypertension and he said the allergist has asked him to decide if he wants to take SCIT or SLIT. He asks you which factors he should consider. What is the BEST answer?
A. Advise him to consider cost, dosing frequency, and route of administration
B. Advise him to consider cost and convenience alone as they are both effective
C. Advise him to tell his allergist he is taking a beta blocker, so SLIT is preferred
7. Emily and her mom come to the pharmacy, and they are very excited. They are considering adopting a dog! Emily has asthma and multiple allergies, and the pediatrician has told them she is probably allergic to or will become allergic to dogs. As Mom chatters, she tells you that the 9-year-old dog, Raven, is an Alaskan Malamute (a breed that has a heavy undercoat) that weighs 95 pounds. She said that a friend told her that if she washes the dog two or three times a month, allergies will not be a problem. She says, “I think I can find time to wash a dog twice a month.” What is the MOST IMPORTANT FACT should you bring to her attention?
A. Before adopting Raven, check with a veterinarian or a breed advocacy group to determine if bathing is a good idea.
B. Bathing a pet two to three times a month is not frequent enough to reduce the allergen load—you have to bathe them two to three times a week.
C. Look for a younger Alaskan Malamute—maybe a puppy—so the dog will get used to being bathed all the time.
8. Adele., who is 7 months pregnant, is experiencing an allergic reaction to a visiting ferret. She asks you to recommend an OTC product to reduce her nasal stuffiness and itchy eyes. Which is the BEST product to recommend?
A. Intranasal cromolyn
B. Oral levocetirizine
C. Oral diphenhydramine
9. Which of the following have similar effectiveness for pet allergies, but different cost effectiveness?
A. Fluticasone and fexofenadine
B. Montelukast and loratadine
C. Levocetirizine and fexofenadine
10. Which medication class should be used for no longer than five days at a time?
A. Decongestants
B. First generation antihistamines
C. Nasal corticosteroids
References
Full List of References
- 2021-2022 APPA National Pet Owners Survey. Accessed January 17, 2022. https://www.americanpetproducts.org/press_industrytrends.asp
- Ownby D, Johnson C. Recent Understandings of Pet Allergies [version 1; peer review: 2 approved]. F1000Research. 2016;5(108)doi:10.12688/f1000research.7044.1
- Perzanowski MS, Rönmark E, Platts-Mills TA, Lundbäck B. Effect of cat and dog ownership on sensitization and development of asthma among preteenage children. Am J Respir Crit Care Med. 2002;166(5):696-702. doi:10.1164/rccm.2201035
- Ownby DR, Johnson CC, Peterson EL. Exposure to dogs and cats in the first year of life and risk of allergic sensitization at 6 to 7 years of age. JAMA. 2002;288(8):963-72. doi:10.1001/jama.288.8.963
- Díaz-Perales A, González-de-Olano D, Pérez-Gordo M, Pastor-Vargas C. Allergy to uncommon pets: new allergies but the same allergens. Front Immunol. 2013;4:492-492. doi:10.3389/fimmu.2013.00492
- Curin M, Hilger C. Allergy to pets and new allergies to uncommon pets. Allergol Select. 2017;1(2):214-221. Published 2017 Aug 4. doi:10.5414/ALX01842E
- Villas F, Compes E, Fernández-Nieto M, Muñoz MP, Bartolome B, de las Heras M. Bird-egg syndrome caused by Agapornis species (lovebird). J Investig Allergol Clin Immunol. 2009;19(1):71-2.
- Quirce S. Asthma in Alergológica-2005. J Investig Allergol Clin Immunol. 2009;19 Suppl 2:14-20.
- Ferret allergies: Are ferrets hypoallergenic animals? Accessed July 12, 2022. https://friendlyferret.com/ferret-hypoallergenic-allergies/
- Are rabbits hypoallergenic? All your questions answered. Hypoallergenic Home. Accessed July 12, 2022. https://hypoallergenichomes.com/hypoallergenic-pets/rabbits/
- Konradsen JR, Fujisawa T, van Hage M, et al. Allergy to furry animals: New insights, diagnostic approaches, and challenges. J Allergy Clin Immunol. Mar 2015;135(3):616-25. doi:10.1016/j.jaci.2014.08.026
- Kang SY, Won HK, Park SY, Lee SM, Lee SP. Prevalence and diagnostic values of laboratory animal allergy among research personnel [published online ahead of print, 2021 Jul 11]. Asian Pac J Allergy Immunol. 2021;10.12932/AP-220321-1094. doi:10.12932/AP-220321-1094
- Grönlund H, Saarne T, Gafvelin G, van Hage M. The major cat allergen, Fel d 1, in diagnosis and therapy. Int Arch Allergy Immunol. 2010;151(4):265-74. doi:10.1159/000250435
- Fernández-Parra B, Bisson C, Vatini S, Conti A, Cisteró Bahima A. Allergy to chinchilla. J Investig Allergol Clin Immunol. 2009;19(4):332-3.
- De las Heras M, Cuesta-Herranz J, Cases B, et al. Occupational asthma caused by gerbil: purification and partial characterization of a new gerbil allergen. Ann Allergy Asthma Immunol. 2010;104(6):540-542.
- De las Heras M, Cuesta J, De Miguel J, et al. Occupational rhinitis and asthma caused by gerbil. J Allergy Clin Immunol. 2002;109(1):S326.
- Hunskaar S, Fosse RT. Allergy to laboratory mice and rats: a review of the pathophysiology, epidemiology and clinical aspects. Lab Anim. 1990;24(4):358-379.
- Sathish JG, Sethu S, Bielsky M-C, et al. Challenges and approaches for the development of safer immunomodulatory biologics. Nat Rev Drug Discov. 2013;12(4):306-324.
- Phipatanakul W. Rodent allergens. Curr Allergy Asthma Rep. 2002;2(5):412-416.
- Gonzáles de Olano D, Pastor Vargas C, Cases Ortega B, et al. Identification of a novel 17-kDa protein as a ferret allergen. Ann Allergy Asthma Immunol.. 2009;103(2):177-178.
- Posthumus J, James HR, Lane CJ, et al. Initial description of pork-cat syndrome in the United States. J Allergy Clin Immunol.. 2013;131(3):923-925.
- FDA Allergen Extract Sublingual Tablet. Cited 21 February 2022 Accessed https://www.fda.gov/vaccines-blood-biologics/allergenics/allergen-extract-sublingual-tablets.
- Jesner S. (2022, June 28). Sublingual immunotherapy faqs. Sublingual Immunotherapy FAQs. Accessed July 8, 2022. https://www.hopkinsmedicine.org/otolaryngology/specialty_areas/sinus_center/sublingual_immunotherapy.html#:~:text=Immunotherapy%20treats%20the%20cause%20of,as%20drops%20under%20the%20tongue.
- Malandain H. IgE antibody in the serum--the main problem is cross-reactivity. Allergy. 2004;59(2):229-230. doi:10.1046/j.1398-9995.2003.00395.x
- Nicholas CE, Wegienka GR, Havstad SL, et al. Dog allergen levels in homes with hypoallergenic compared with nonhypoallergenic dogs. Am J Rhinol Allergy. 2011;25(4):252-6. doi: 10.2500/ajra.2011.25.3606
- Dance A. The race to deliver the hypoallergenic cat. Nature. 2020;588(7836):S7-S9. doi:10.1038/d41586-020-02779-3
- Hypoallergenic cats. Blue Cross. (n.d.). Accessed July 8, 2022. https://www.bluecross.org.uk/advice/cat/hypoallergenic cats#:~:text=Despite%20popular%20belief%2C%20hypoallergenic%20cats,how%20much%20protein%20they%20produce
- Skin prick tests. FoodAllergy.org. Accessed August 1, 2022. Skin Prick Tests - FoodAllergy.org
- de Vos G. Skin testing versus serum-specific IgE testing: which is better for diagnosing aeroallergen sensitization and predicting clinical allergy?. Curr Allergy Asthma Rep. 2014;14(5):430. doi:10.1007/s11882-014-0430-z
- Hodson T, Custovic A, Simpson A, Chapman M, Woodcock A, Green R. Washing the dog reduces dog allergen levels, but the dog needs to be washed twice a week. J Allergy Clin Immunol. Apr 1999;103(4):581-5. doi:10.1016/s0091-6749(99)70227-7
- Latz K. How Often Should You Bathe Your Dog? Accessed July 13, 2022. https://www.akc.org/expert-advice/health/how-often-should-you-wash-your-dog/
- Aalberse RC. Mammalian airborne allergens. Chem Immunol Allergy. 2014;100:243-247. doi:10.1159/000358862
- Wood RA, Johnson EF, Van Natta ML, Chen PH, Eggleston PA. A placebo-controlled trial of a HEPA air cleaner in the treatment of cat allergy. Am J Respir Crit Care Med. 1998;158(1):115-120. doi:10.1164/ajrccm.158.1.9712110
- Discover ProPlan LiveClear Allergen Reducing Cat Food. Purina. Accessed July 12, 2022. https://www.purina.com/pro-plan/cats/liveclear-cat-allergen-reducing-food
- Scolaro KL. Chapter 11: Colds and Allergy. Handbook of Nonprescription Drugs: An Interactive Approach to Self-Care, 20th Edition.
- Product Information: SINGULAIR(R) oral tablets, oral chewable tablets, oral granules, montelukast sodium oral tablets, oral chewable tablets, oral granules. Merck & Co (Per FDA); 2012.
- Clark J, White ND. Immunotherapy for Cat Allergies: A Potential Strategy to Scratch Back. Am J Lifestyle Med. 2017;11(4):310-313. doi:10.1177/1559827617701389
- Allergenics. U.S. Food and Drug Administration. Accessed July 29, 2022. https://www.fda.gov/vaccines-blood-biologics/allergenics
- Sublingual Immunotherapy. Johns Hopkins Medicine. Accessed July 13, 2022. https://www.hopkinsmedicine.org/otolaryngology/specialty_areas/sinus_center/sublingual_immunotherapy.html#:~:text=Immunotherapy%20treats%20the%20cause%20of,as%20drops%20under%20the%20tongue.
- Abramson MJ, Puy RM, Weiner JM. Injection allergen immunotherapy for asthma. Cochrane Database Syst Rev. 2010;(8):Cd001186. doi:10.1002/14651858.CD001186.pub2
- Aasbjerg K, Torp-Pedersen C, Backer V. Specific immunotherapy can greatly reduce the need for systemic steroids in allergic rhinitis. Allergy. 2012;67(11):1423-9. doi:10.1111/all.12023
- Ling M, Long AA. Pet dander and difficult-to-control asthma: therapeutic options. Allergy Asthma Proc. 2010;31:385-391.
- Cox L, Nelson H, Lockey R, et al. Allergen immunotherapy: a practice parameter third update. J Allergy Clin Immunol. 2011;127(1 Suppl):S1-55. doi:10.1016/j.jaci.2010.09.034
- Goodman MJ, Jhaveri M, Saverno K, Meyer K, Nightengale B. Cost-effectiveness of second-generation antihistamines and montelukast in relieving allergic rhinitis nasal symptoms. Am Health Drug Benefits. 2008;1(8):26-34.
- Aboshady OA, Elghanam KM. Sublingual immunotherapy in allergic rhinitis: efficacy, safety, adherence and guidelines. Clin Exp Otorhinolaryngol. 2014 Dec;7(4):241-9. doi: 10.3342/ceo.2014.7.4.241.
- Hardin FM, Eskander PN, Franzese C. Cost-effective Analysis of Subcutaneous vs Sublingual Immunotherapy From the Payor's Perspective. OTO Open. 2021 Oct 25;5(4):2473974X211052955. doi: 10.1177/2473974X211052955.