Learning Objectives
After completing this knowledge-based continuing education activity, pharmacists will be able to
- RECALL symptoms associated with dengue fever and malaria
- DESCRIBE emerging information about dengue and malaria vaccines
- ASSOCIATE dengue fever and malaria vaccines for specific patients
After completing this knowledge-based continuing education activity, pharmacy technicians will be able to
- RECALL symptoms associated with dengue fever and malaria
- DESCRIBE emerging information about dengue and malaria vaccines
- MATCH dengue fever and malaria vaccines by storage requirements

Release Date: February 28, 2026
Expiration Date: February 28, 2029
Course Fee
Pharmacists: $4
Pharmacy Technicians: $2
There is no grant funding for this CE activity
ACPE UANs
Pharmacist: 0009-0000-26-007-H06-P
Pharmacy Technician: 0009-0000-26-007-H06-T
Session Codes
Pharmacist: 23YC02-MTX44
Pharmacy Technician: 23YC02-XTM62
Accreditation Hours
0.5 hours of CE
Accreditation Statements
| The University of Connecticut School of Pharmacy is accredited by the Accreditation Council for Pharmacy Education as a provider of continuing pharmacy education. Statements of credit for the online activity ACPE UAN 0009-0000-26-007-H06-P/T will be awarded when the post test and evaluation have been completed and passed with a 70% or better. Your CE credits will be uploaded to your CPE monitor profile within 2 weeks of completion of the program. |  |
Disclosure of Discussions of Off-label and Investigational Drug Use
The material presented here does not necessarily reflect the views of The University of Connecticut School of Pharmacy or its co-sponsor affiliates. These materials may discuss uses and dosages for therapeutic products, processes, procedures and inferred diagnoses that have not been approved by the United States Food and Drug Administration. A qualified health care professional should be consulted before using any therapeutic product discussed. All readers and continuing education participants should verify all information and data before treating patients or employing any therapies described in this continuing education activity.
Faculty
Jessica Bylyku, BS
PharmD Candidate 2024
UConn School of Pharmacy
Storrs, CT
Kelsey Giara, PharmD
Freelance Medical Writer
Pelham, NH
Melody White
PharmD Candidate 2025
UConn School of Pharmacy
Storrs, CT
Faculty Disclosure
In accordance with the Accreditation Council for Pharmacy Education (ACPE) Criteria for Quality and Interpretive Guidelines, The University of Connecticut School of Pharmacy requires that faculty disclose any relationship that the faculty may have with commercial entities whose products or services may be mentioned in the activity.
Dr. Giara, Jessica Bykylu and Melody White do not have any relationships with ineligible companies and therefore have nothing to disclose.
ABSTRACT
Malaria and dengue (pronounced deng-ee) fever are not new diseases, but given the emergence of new vaccines, it is critical that pharmacists and pharmacy technicians increase their familiarity with them. These illnesses are both transmitted by mosquitos, but malaria is caused by Plasmodium parasites while dengue fever is a viral disease caused by dengue virus. The United States is not a malaria- or dengue-endemic country, but travel to other countries puts people at risk of these conditions. Pharmacy teams should be prepared to identify potential cases and refer patients for appropriate vaccination or treatment when appropriate.
CONTENT
Content
INTRODUCTION
Malaria cases in 2020 totaled an estimated 241 million, leading to more than 600,000 deaths, mostly in Africa.1 Direct costs of malaria prevention and treatment in the United States (U.S.) total about $12 billion annually, excluding the toll it takes on affected individuals and their families.1 The World Health Organization (WHO) reports that between 100 to 400 million people are infected with dengue fever each year.2 About 80% of cases are mild and asymptomatic, but dengue fever can progress to “severe dengue,” which is classified as a medical emergency requiring immediate medical care.2,3
Mosquitos, Malaria, and Dengue – Oh My!
Plasmodium parasites—common to tropical areas (e.g., Africa, South America, the Caribbean Islands, South Asia)—cause malaria.1 Most commonly, malaria is transmitted through the bite of infected mosquitoes, specifically the Anopheles species, during local outbreaks. There is also a term coined “airport malaria,” describing disease that is transported from an infected country to a non-infected country.4 Congenital malaria occurs when mothers infected with the disease transmit parasites to the child during pregnancy or birth.4 Although rare, prompt diagnosis is crucial to ensure infected neonates and infants survive. Transfusion-transmitted malaria is also possible where blood recipients can be infected with malaria accidently. There are no approved tests to screen blood donations for malaria, only questioning of prospective donors.4 Although rare in the U.S., complications are severe and organizations should take action to prevent potentially-infected individuals from donating.
Patients with malaria generally present with fever, chills/sweating, headache, and weakness within 10 to 15 days of infection.5 Diarrhea, abdominal pain, and cough are also possible. As malaria progresses, patients develop a classic paroxysm (i.e., symptoms that come and go) comprising three stages6:
- 15-to-60-minute cold stage (shivering and feeling cold)
- 2-to-6-hour hot stage (fevers up to nearly 106°F; flushed, dry skin; and often headache, nausea, and vomiting)
- 2-to-4-hour sweating stage (rapid drop in fever and sweating)
Missed or delayed malaria diagnosis can lead to potentially fatal complicated disease manifesting as severe anemia, renal failure, altered consciousness, and multisystem organ failure.6 Clinicians diagnose malaria via a blood smear test. Although rapid and polymerase chain reaction (PCR) tests are available, medical professionals confirm diagnosis through microscopic blood smear examination.7
Dengue fever is a viral disease caused by mosquitos—mainly females from the Aedes aegypti and Ae. albopictus species—carrying dengue virus (also known as DENV).2 Four DENV serotypes exist, so it is possible to contract the disease four times. The virus can be transmitted through mosquito bite, from pregnant mother to child, and via infected blood products/organ donations and infusions. Transovarial transmission within mosquitoes (from parent to offspring) has also been noted.2
Most dengue cases are asymptomatic or mild and fatalities are rare, but increasing severity can be life-threatening.2,3 Providers should suspect dengue when a high fever (104°F or greater) is accompanied by any two of the following symptoms2,3:
- severe headache
- pain behind the eyes
- muscle/joint/bone pain
- nausea/vomiting
- swollen glands
- rash
This febrile phase lasts about 2 to 7 days, and most people recover after about a week.2,3 Severe dengue is a potentially fatal complication due to plasma leakage, fluid accumulation, respiratory distress, severe bleeding, or organ impairment.2 Patients are at risk of severe dengue symptoms about 3 to 7 days after initial symptoms appear.2 As fever drops to below 100°F, patients enter a “critical phase” for 24 to 48 hours. Warning signs to watch for during the critical phase include2
- severe abdominal pain
- rapid breathing
- blood in vomit, stool, gums, or nose
- persistent vomiting
- restlessness/fatigue
Clinicians use commercially available PCR or rapid diagnostic tests to confirm dengue diagnosis.2 Enzyme-linked immunosorbent assays are also available to confirm active or previous infections.
Global Implications
Beyond clinical symptoms, malaria and dengue fever inflict social and financial loss for diagnosed individuals and the countries tasked with treating affected populations. Some examples of the indirect burden of these mosquito-borne diseases include1
- expenses for traveling and receiving treatment
- absences from work/school
- burial expenses in cases of death
- purchases of medication and supplies
- public health interventions (e.g., insecticide spraying, bed nets)
- opportunity loss for tourism
Populations at increased risk of contracting malaria include infants, children younger than 5 years, pregnant women, immunosuppressed patients, and migrant workers or traveling populations.5 There is also concern that certain mosquitoes are resistant to insecticide, and by migrating throughout the world they can spread malaria to urban populations.8 Researchers have identified Anopheles gambiae mosquitoes, originally found in India and Iran, as insecticide-resistant. These are projected to put nearly 126 million people in African cities at risk for contracting malaria.8
Populations most vulnerable to contracting dengue fever include pregnant women and children.3 Many asymptomatic or mild dengue cases go unreported. WHO reports most of the dengue burden occurs in Asia, and the number of cases has steadily increased to just over 5 million in 2019.2
PREVENTION AND TREATMENT
Following prevention and treatment guidelines are crucial to lower transmission rates of dengue fever and malaria.
Dengue Fever
WHO states that countries should be aware of community mosquito presence and develop active mosquito and virus surveillance to prevent further disease spread.2 They should also remain knowledgeable about the number of infected individuals.
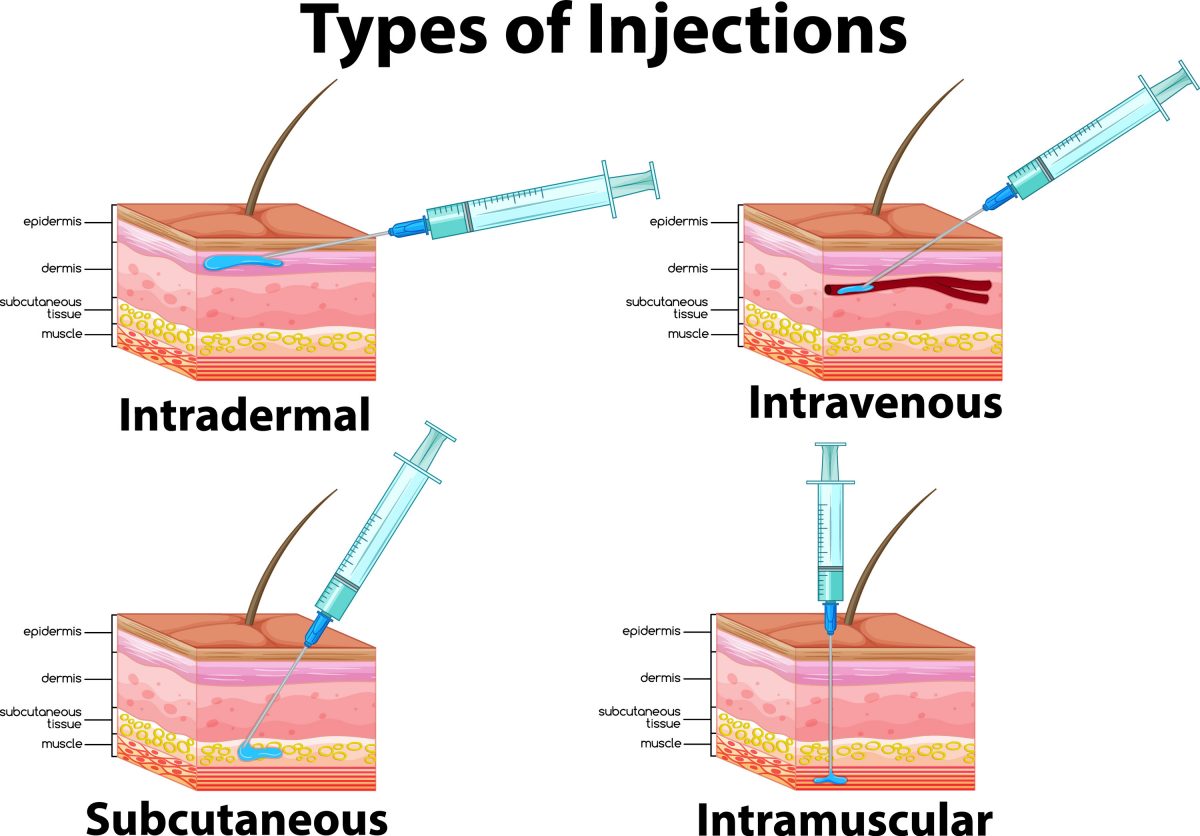
The dengue vaccine (Dengvaxia) has been licensed in other countries since 2015, but the U.S. Food and Drug Administration (FDA) approved the vaccine in 2019.2 WHO recommends people aged 9 to 45 years be vaccinated, but Dengvaxia is only FDA approved for patients 9 to 16 years old with a history of previous infection who live in high-risk areas. As a live-attenuated vaccine, it is contraindicated in individuals with severe immunodeficiency.2 Children receiving Dengvaxia need a 3-dose series administered subcutaneously with doses separated by 6 months.9 Providers should store the vaccine in the refrigerator.10 After reconstitution, it should be administered immediately or stored in the refrigerator and used within thirty minutes.
WHO and the FDA only recommend Dengvaxia for patients with a history of dengue virus infection.10,11 This is based on clinical trial evidence that the vaccine is efficacious and safe in patients with a history of previous DENV infection because a subsequent infection is more serious and life-threatening than the first.11 They also advise countries using the vaccine to control viral spread to implement pre-vaccination screening to confirm previous infection.
As no dengue-specific treatment is available, providers should treat infected patients symptomatically with acetaminophen, rest, and fluids.2 Patients with dengue fever should avoid non-steroidal anti-inflammatory drugs (e.g., ibuprofen, aspirin) because they thin the blood. Given the risk of hemorrhage in this disease, blood thinners may exacerbate the problem.2
Malaria
WHO recommends that countries engage in vector control and surveillance for the spread of malarial disease.5 Malaria vaccines have been in development for decades, but no malaria vaccine is available in the U.S.12 In 2021, however, WHO recommended a new malaria vaccine (Mosquirix) for children aged older than 5 months who live in areas with moderate to high transmission of P. falciparum.13 The vaccine is only recommended for children as malaria is one of the main killers of children younger than 5 years in countries with moderate or high rates of malaria.14 WHO also recommends giving the vaccine seasonally in countries where malaria transmission is high during certain seasons.13
Initial Mosquirix pilot studies are ongoing, and more widespread vaccine rollout is expected in 2023. For now, people in the U.S. traveling to malaria-endemic countries continue to use oral medications as chemoprophylaxis (i.e., to prevent the disease), including atovaquone/proguanil, chloroquine, doxycycline, mefloquine, primaquine, and tafenoquine.15
Clinicians administer Mosquirix as a 4-dose schedule.16 The vaccine’s adverse effects are pain and swelling at the injection site and fever.17 Providers should store the vaccine in the refrigerator. After reconstitution it should be administered immediately or stored in the refrigerator and used within 6 hours.16
Malaria treatment involves the use of antimalarial drugs based on four main factors15:
- Infection severity: Malaria infection is either considered uncomplicated (effectively treated with oral antimalarials) or severe (requiring aggressive intravenous antimalarial therapy).
- Infecting Plasmodium species: P. falciparum and P. knowlesi infections can cause rapidly progressive severe illness or death, necessitating urgent therapy initiation, while other species are less likely to cause severe disease. P. vivax and P. ovale infections also require treatment for hypnozoites (parasites that lay dormant in the liver and then re-awaken to become active infectants).
- Drug susceptibility: In addition to disease severity differences, Plasmodium species also have different drug susceptibilities, so providers select an antimalarial therapy based on the species of the infecting parasite. If the species cannot be determined, patients must initiate antimalarial treatment against chloroquine-resistant P. falciparum as soon as possible.
- Previous antimalarial use: Patients using antimalarial medication as chemoprophylaxis, should not receive that same drug or drug combination to treat malaria infection unless no other options are available.
CONCLUSION
Pharmacists and pharmacy technicians should be familiar with the signs and symptoms of malaria and dengue fever to inform patients when these conditions are suspected and about their appropriate treatment. Pharmacy teams who suspect a case of malaria or dengue fever should refer patients for medical attention and contact their local or state health department.
Pharmacist Post Test (for viewing only)
PHARMACIST POSTTEST
Learning Objectives
● RECALL symptoms associated with dengue fever and malaria
● DESCRIBE emerging information about dengue and malaria vaccines
● OPTIMIZE dengue fever and malaria vaccines for specific patients
1. Which of the following is TRUE?
A. Dengue fever symptoms are always severe and most patients die
B. Malaria presents as a cold stage, hot stage, and sweating stage
C. Pain behind the eyes is a warning sign for malaria
2. Which of the following is TRUE regarding the malaria vaccine?
A. WHO recommends it for children 5 months and older who live in endemic areas
B. It is FDA approved for patients 9 to 16 years old with a history of previous infection
C. Clinicians administer it as a 3-dose series with each dose separated by 6 months
3. A patient comes to the pharmacy indicating she and her family are being transferred to a country where dengue is common. She wants to have her three children who are ages 3, 5, and 7 vaccinated for dengue before they move. What is the BEST thing to tell her?
A. We should schedule your children to be vaccinated about six weeks before you plan to move so they develop antibodies before you actually relocate.
B. We only vaccinate children who have already had dengue because a second infections is more serious and life-threatening than the first.
C. Wait until you arrive in the country because they will want to do pre-vaccination screening to confirm your children have not been infected previously.
Pharmacy Technician Post Test (for viewing only)
PHARMACY TECHNICIAN POSTTEST
Learning Objectives
● RECALL symptoms associated with dengue fever and malaria
● DESCRIBE emerging information about dengue and malaria vaccines
● CLASSIFY dengue fever and malaria vaccines by storage requirements
1. Which of the following is TRUE?
A. Dengue fever symptoms are always severe and most patients die
B. Malaria presents as a cold stage, hot stage, and sweating stage
C. Pain behind the eyes is a warning sign for malaria
2. Which of the following is TRUE regarding the malaria vaccine?
A. WHO recommends it for children 5 months and older who live in endemic areas
B. It is FDA approved for patients 9 to 16 years old with a history of previous infection
C. Clinicians administer it as a 3-dose series with each dose separated by 6 months
3. A patient at your pharmacy is receiving the dengue fever vaccine. The patient’s mother asks you if they can use the restroom before the pharmacist administers the vaccine. You look over to see that the pharmacist has just finished reconstituting Dengvaxia for this patient. Which of the following is the BEST response?
A. Advise the mother not to leave the pharmacy waiting area, as the pharmacist needs to administer this vaccine immediately or it will expire
B. Advise the mother to take her daughter to the restroom, and the pharmacist can administer this vaccine within 6 hours as long as it’s refrigerated
C. Advise the mother to take her daughter to the restroom but return within 30 minutes, and ensure the pharmacist refrigerates the reconstituted vaccine
References
Full List of References
REFERENCES
- Centers for Disease Control and Prevention. Malaria’s Impact Worldwide. Updated December 16, 2021. Accessed November 30, 2022. https://www.cdc.gov/malaria/malaria_worldwide/impact.html
- World Health Organization. Dengue and severe dengue. Updated January 10, 2022. Accessed November 1, 2022. https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue
- Centers for Disease Control and Prevention. Dengue. Updated August 3, 2022. Accessed November 1, 2022. https://www.cdc.gov/dengue/index.html
- Centers for Disease Control and Prevention. Parasites – Malaria. Updated August 19, 2022. Accessed November 1, 2022. https://www.cdc.gov/parasites/malaria/index.html
- World Health Organization. Malaria. Updated July 26, 2022. Accessed November 1, 2022. https://www.who.int/news-room/fact-sheets/detail/malaria
- Crutcher JM, Hoffman SL. Chapter 83: Malaria. In: Baron S, ed. Medical Microbiology. 4th ed. University of Texas Medical Branch at Galveston; 1996. Accessed November 1, 2022. https://www.ncbi.nlm.nih.gov/books/NBK8584/
- Stanford Health Care. Malaria diagnosis. Accessed November 1, 2022. https://stanfordhealthcare.org/medical-conditions/primary-care/malaria/diagnosis.html
- American Society of Tropical Medicine and Hygiene. Invasive mosquitos – Anopheles stephensi in Ethiopia. November 1, 2022. Accessed November 30, 2022. https://astmhpressroom.wordpress.com/annual-meeting-2022/anopheles-stephensi-in-ethiopia/
- Centers for Disease Control and Prevention. Dengue Vaccine VIS. Updated December 17, 2021. Accessed November 29, 2022. https://www.cdc.gov/vaccines/hcp/vis/vis-statements/dengue.html
- Dengvaxia [prescribing information]. Sanofi Pasteur; 2019.
- Ask the Experts: Dengue. Immunize.org. Updated February 16, 2022. Accessed November 21, 2022. https://www.immunize.org/askexperts/experts_dengue.asp
- Centers for Disease Control and Prevention. Malaria: Vaccines. Updated October 7, 2021. Accessed December 2, 2022. https://www.cdc.gov/malaria/malaria_worldwide/reduction/vaccine.html
- Q&A on RTS,S malaria vaccine. World Health Organization. Updated April 21, 2022. Accessed November 30, 2022. https://www.who.int/news-room/questions-and-answers/item/q-a-on-rts-s-malaria-vaccine
- UNICEF. Millions more children to benefit from malaria vaccine as UNICEF secures supply. August 16, 2022. Accessed November 22, 2022. https://www.unicef.org/press-releases/millions-more-children-benefit-malaria-vaccine-unicef-secures-supply
- Centers for Disease Control and Prevention. Treatment of Malaria: Guidelines for Clinicians (United States). Updated September 30, 2022. Accessed November 30, 2022 https://www.cdc.gov/malaria/diagnosis_treatment/clinicians1.html
- Mosquirix (Product Information). European Medicines Agency. Updated January 8, 2022. Accessed November 30, 2022. https://www.ema.europa.eu/en/documents/outside-eu-product-information/mosquirix-product-information_en.pdf
- World Health Organization. Malaria: The malaria vaccine implementation programme (MVIP). March 2, 2020. Accessed November 30, 2022. https://www.who.int/news-room/questions-and-answers/item/malaria-vaccine-implementation-programme