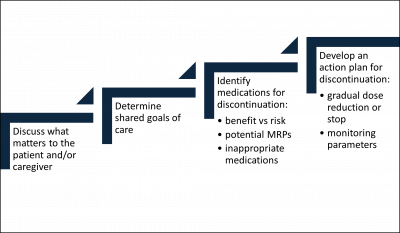INTRODUCTION
The number of older adults (aged 65 years and older) is expected to exceed 1.5 billion by 2050.1 Multimorbidity is expected to affect 65% of older adults aged 65 to 84 years and up to 82% of those aged 85 years or older.1 With multimorbidity, the number of medications taken by the vulnerable older adult population increases. Along with drug-disease and drug-drug interactions, age-related physiological changes that alter pharmacokinetics and pharmacodynamics increase the risk of adverse drug events (ADEs).2
Throughout this continuing education activity, consider a hypothetical patient, Ms. Polly, who is an 85-year-old female. Enrolled in a Program of All-Inclusive Care for the Elderly (PACE), she has been able to live safely at home. Table 1 lists her current medication regimen.
| Table 1. Medication Regimen |
| Acetaminophen/diphenhydramine PM 25/500 mg, 2 tablets nightly PRN for insomnia
Alendronate 70 mg weekly for osteoporosis
Amlodipine 10 mg daily for hypertension
Aspirin 81 mg daily for heart attack prevention
Cetirizine 10 mg daily for allergies
Donepezil 10 mg daily for Alzheimer’s disease
Fluticasone nasal spray 2 sprays in each nostril daily for allergies
Furosemide 20 mg daily for peripheral edema
Gabapentin 300 mg twice daily for neuropathic pain
Glipizide ER 5 mg daily for pre-diabetes
Ibuprofen 200 mg three times daily for pain
Omega-3 Fish Oil 1000 mg twice daily for hyperlipidemia
Oxybutynin 5 mg twice daily for overactive bladder
Pantoprazole 40 mg daily for GERD
Paroxetine 30 mg daily for depression
Potassium chloride 10 mEq daily for potassium deficiency
Pravastatin 40 mg daily for hyperlipidemia
Propranolol 40 mg twice daily for hypertension
Senna-docusate 8.6-50 mg, 2 tablets daily for constipation |
ABBREVIATIONS: GERD = gastroesophageal reflux disease; mEq = milliequivalent; mg = milligrams; PRN = as needed
Ms. Polly’s healthcare needs have become more complex with her increasing confusion, declining ability to perform activities of daily living, and a recent fall. Her daughter continues to be her main caregiver and has requested more help from PACE; thus, the prescriber has reached out to the clinical pharmacist for assistance. This CE activity will review best practices for deprescribing with a focus on why, who, when, what, and how to safely deprescribe medications in the vulnerable older adult population.
WHY, WHO, AND WHEN TO DEPRESCRIBE
WHY: Older Adults Have Specific Needs
Since older adults have differing health and functional statuses, prescribers must individualize medication selection for these patients. Prescribing the right medication and dose for the right indication at the right time while providing benefit and avoiding ADEs is challenging. Comorbidities and complexity of medication regimens may result in suboptimal medication use. Suboptimal medication use comprises (1) underprescribing, (2) polypharmacy or overuse, and (3) high-risk prescribing, such as potentially inappropriate medications (PIMs), prescribing cascades, and anticholinergic and sedative effects.2
Estimates suggest that as many as 65% of older adults in the United States are exposed to polypharmacy and 29% to PIMs.3,4 Polypharmacy is generally defined as the use of five or more medications and often occurs because of a prescribing cascade, which means the culprit medication causes an ADE that is mistaken for a new medical condition for which a subsequent medication is initiated as treatment. “Hyperpolypharmacy” or “excessive polypharmacy” is generally considered the use of 10 or more medications.5 Polypharmacy is associated with undesirable outcomes: frailty, cognitive decline, functional decline, falls, fractures, emergency department (ED) visits, hospitalizations, and mortality.3,5,6
Evidence suggests that older age and indicators of poor health (i.e., multiple chronic conditions, cognitive decline, frailty) are risk factors for polypharmacy. The number of medications in a patient’s regimen may be the single most important predictor of ADEs.6 The likelihood of taking inappropriate medication increases with the number of drugs prescribed. Experts consider medications inappropriate in older adults when they pose individual or cumulative adverse event risk.
The Choosing Wisely Campaign brought awareness and encouraged conversations between healthcare providers and patients about medication overuse and potentially doing more harm than good within healthcare. This partnership between the American Board of Internal Medicine (ABIM) Foundation and specialty societies was formed in 2012. At the time, the healthcare field and society generally thought that more care was better, and patients tended to perceive prescribers as ignoring their patients’ interests if they did not prescribe certain medications.7
Understanding suboptimal prescribing allows healthcare providers to mitigate the risk of poor health outcomes in vulnerable older adults. Deprescribing is the process of healthcare professional-supervised medication withdrawal with the goal of managing polypharmacy and improving outcomes.8 Pharmacists review medication regimens comprehensively to identify and consider discontinuation of high-risk medications and those that cause more harm than benefit. Deprescribing encourages a proactive and systematic approach to mitigate potential medication-related problems (MRPs). This approach is preferable over a reactive approach (i.e., discontinuing an offending medication after an ADE occurs).
PAUSE AND PONDER: Why should healthcare providers re-evaluate medications periodically?
According to the World Health Organization, the global ADE-related mortality rate increased approximately 3.3-fold from 2001 to 2019, with the highest rates occurring in those aged 75 years and older.9 A systematic review of 14 hospital-based observational studies in older adults found an ADE to occur more often with an increased number of medications.10 More than 50% of ADEs could be prevented with safe prescribing practices.5 Safe medication use requires that healthcare providers ensure regimen appropriateness, considering treatment initiation, treatment optimization, and cessation of unnecessary or inappropriate medications.
WHO: The Age-Friendly Movement
The Institute for Healthcare Improvement in partnership with The John A. Hartford Foundation, American Hospital Association, and the Catholic Health Association developed the Age-Friendly Health Systems initiative to improve the care of older adults and the lives of caregivers. This framework—the Age-Friendly 4Ms, depicted in Figure 1—is a patient-centered approach to medication optimization that addresses polypharmacy and deprescribing. Conversations with healthcare providers can be re-framed to discuss “What Matters” to the patients and caregivers. Medications impact all the “M” domains: What Matters, Mentation and Mobility.11,12
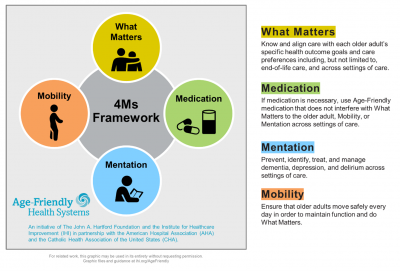
Figure 1. Age-Friendly Health Systems 4Ms Framework11
Reasons for deprescribing may vary from patient to patient. When reviewing common goals for deprescribing, consider the perspectives of the patient, the caregiver, and the healthcare team. Identify which reasons for deprescribing may be most relevant or meaningful to each party involved in the patient’s care. Examples include
- reducing medication regimen complexity and pill burden to improve adherence
- reducing anticholinergic burden to improve and/or preserve cognitive function
- reducing fall and fracture risk
- reducing hospitalizations
- reducing mortality
- reducing costs
These deprescribing goals improve patients’ quality of life. Deprescribing considers not only single medications but also the aggregate interaction risk from multiple medications.13
Deprescribing often presents challenges due to barriers faced by healthcare providers and patients. Healthcare providers’ may have limited time and lack of training which serves as the most significant barriers. Patient-related barriers to deprescribing include satisfaction with medications, reluctance, resistance to change, and opposition to alternative treatments. Despite these challenges, many barriers can be addressed through education, communication, and shared decision-making.
Although many patients express not wanting to take medications, their beliefs and acceptance of polypharmacy and discontinuing medications is poorly understood. To further understand patients’ views, preferences and willingness, researchers from the University of South Australia and University of Sydney developed the Patients’ Attitudes Towards Deprescribing (PATD) questionnaires, including the revised PATD for older adults and caregivers and the revised PATD for cognitively impaired individuals (rPATDcog).14-16
A population-based survey of Medicare beneficiaries 65 years and older using PATD questions found most older adults are open to having one or more medications deprescribed and two-thirds want to reduce the number of medications taken.17 A multi-center cross-sectional survey found older adults and caregivers are open to deprescribing, with more than 90% willing to discontinue a medication if a healthcare provider initiates the conversation.18
While primarily used for research, providers may use the revised PATD questionnaire clinically to target patients and caregivers who are willing to discontinue medications. The original work is available here: https://www.australiandeprescribingnetwork.com.au/925-2/.16 Understanding attitudes and willingness may enhance shared decision-making conversations about deprescribing.
PAUSE AND PONDER: How do you determine which medications to deprescribe?
WHEN TO DEPRESCRIBE
Older adults are often high-risk for potential MRPs due to pharmacokinetic (the body’s effect on a medication) and pharmacodynamic (a medication’s effect on the body) changes. However, advanced age is not an independent risk factor for MRPs. Elements of high-risk patients who may benefit from deprescribing include the following19:
- Polypharmacy: Use of multiple medications is the strongest risk factor for MRPs. Table 2 lists common types of potential MRPs.
- Multimorbidity: Having multiple chronic conditions increases the risk of drug-disease interactions.
- Renal impairment: Reduced renal function increases ADE risk.
- Transitions of care: Medication management by multiple prescribers can result in miscommunication and medication discrepancies, such as inadvertent continuation.
- Nonadherence: Patients may be nonadherent to their medication regimens due to experiencing real or perceived adverse effects and/or lack of benefit, complexity, burden, and cost.
- Limited life expectancy/end of life care: Goals of care change from slowing disease progression and prolonging life to improving quality of life. Patients may no longer benefit from preventive medications and use certain medications for symptomatic management.
- Frailty and dementia: Frailty—an age-related syndrome of physiologic decline—is associated with increased adverse events and risk of harm, such as falls, which can be detrimental. ADEs may worsen or exacerbate these syndromes (i.e., fatigue or cognitive impairment).
| Table 2. Types of Potential Medication-Related Problems |
| Adverse Drug Events
Adverse Drug Interactions
Excessive Duration
Inappropriate/Unnecessary Medications
Ineffective Therapy
No Indication
Nonadherence
Suboptimal Dosing
Supratherapeutic Dosing |
Aging is not an identical experience for every person, so frailty—declining mental and physical resilience, or the ability to bounce back and recover from events like illness and injury—is a more appropriate measure of health status than age alone. Frailty has been associated with poor patient outcomes. The Edmonton Frail Scale is a validated, reliable, and multidimensional screening tool for all healthcare settings. The free tool with a training course is available here: https://edmontonfrailscale.org/. It assesses nine domains: cognition, general health status, functional independence, social support, medication use, nutrition, mood, continence, and functional performance. The highest level of frailty is a score of 17.20 Assessing frailty severity can help with shared decision-making and determining goals of care.
PAUSE AND PONDER: How can you use prescribing clinical tools for deprescribing?
WHAT TO DEPRESCRIBE
While few studies have measured the clinical implications of underprescribing, evidence has shown an association between polypharmacy and PIMs. A systematic review of 38 clinical trials found that deprescribing interventions in community-dwelling older adults (aged 65 years and older) may provide small reductions in use of PIMs and mortality.6 Prescribers and pharmacists should periodically reevaluate any medication initially prescribed for a clinical indication since benefits, risks, conditions, and comorbidities change over time, which may result in inappropriate use. Medication review should consider the following factors: goals of care, remaining life expectancy, treatment targets, time until benefit, number needed to treat (NNT, a statistical measure that indicates how many patients need to be treated with a specific intervention to prevent one additional adverse outcome), number needed to harm, and ADEs.21
Collaborative approaches and numerous clinical application tools highlighted below are available for safe prescribing and to reduce medication overuse. Focusing on medications and potential ADE risk, healthcare providers can employ these clinical tools to identify medications for potential deprescribing.
Risk-Benefit Analysis
Older adults are vulnerable to ADEs associated with “high-risk” single medications and medication classes, including anticoagulants, benzodiazepines, cardiovascular agents, digoxin, hypoglycemics, nonsteroidal anti-inflammatory drugs (NSAIDs), opioids, psychotropics, and those with anticholinergic effects. “High-risk” medication combinations—for example, angiotensin-converting enzyme inhibitors, diuretics, and NSAIDs in patients with chronic kidney disease (known as the “triple whammy”)—augment ADE risk.13
In a “Less is More” special communication about polypharmacy and deprescribing, experts proposed grouping medications into two categories for older adults to determine if the likely benefit outweighs potential risk/harm, while noting that medications may fall into both categories.13
“Disease and/or symptom control medications” are used to manage active disease and symptoms and help maintain quality of life.13 Discontinuing these medications could result in symptom return or functional decline due to disease worsening. Examples include analgesics, anti-anginals, anti-heart failure medications, and levothyroxine. Dose reduction of disease/symptom control drugs may be appropriate if symptoms are mild or intermittent, but these should be discontinued if altogether ineffective. Notably, aggressive treatment may be inappropriate for hypertension or diabetes management in some older adults, as this could increase the risks of hypotension or hypoglycemia, respectively.13
Preventive medications are intended to prevent future morbid events.13 Their use should be guided by assessment of absolute risks and benefits of treatment for individual patients, taking into consideration the time required for benefit (known as "time to benefit"), patient preferences, and estimated life expectancy. For example, osteoporosis guidelines recommend a “drug holiday” (temporary discontinuation of therapy) from bisphosphonates depending on fracture risk after five years of treatment to mitigate risk of atypical femoral fractures and osteonecrosis of the jaw. Despite discontinuation, bisphosphonates’ anti-fracture effects persist because this medication class has long half-lives and remain stored in bone for up to 10 years. Another example is statins, which are commonly used for primary prevention of cardiovascular events. Evidence suggests that statin discontinuation after eight years does not increase cardiovascular event risk.13
Choosing Wisely Campaign
The American Board of Internal Medicine’s global Choosing Wisely (CW) Campaign encourages patient engagement and healthcare provider conversation to choose care that is evidence-based, necessary, and free from harm. Since 2012, a compilation of more than 600 evidence-based statements from approximately 80 healthcare organizations advise healthcare providers, patients, and caregivers about optimization, appropriateness, and avoidance of unnecessary medical testing and treatments, available at https://www.choosingwisely.org/.7,22,23,24
The American Society of Consultant Pharmacists (ASCP) convened a task force in 2018 under the CW campaign to further help pharmacists initiate and implement deprescribing. The task force reinforced deprescribing in older adults by reviewing and providing evidence-based references. ASCP’s guidance provides ten statements focused on avoiding DDIs and prescribing cascades and encouraged medication reviews to mitigate potential ADEs25:
- DO NOT initiate medications to treat new and emerging symptoms without first ascertaining that the new symptom is not an ADE related to an already prescribed medication.
- DO NOT continue medications at care transitions without reviewing and reconciling to verify accurate and complete medication lists in concert with current medical problems.
- DO NOT recommend highly anticholinergic medications in older adults without first considering safer alternatives or non-pharmacologic measures.
- DO NOT use anticholinergics concomitantly with cholinesterase inhibitors for dementia.
- DO NOT use two or more medications known to increase bleeding risk (e.g., antiplatelets, direct oral anticoagulants [DOACs], warfarin, aspirin, NSAIDs, corticosteroids, selective serotonin reuptake inhibitors), without evaluating the potential risks and benefits.
- DO NOT prescribe or routinely continue medications for older adults with limited life expectancy without consideration to individual goals of care, presence of comorbidities, and time to benefit for preventive medications.
- DO NOT use three or more central nervous system-active medications (e.g., antidepressants, antipsychotics, benzodiazepines, antiepileptics, Z-drugs, opioids, gabapentinoids).
- DO NOT combine opioids with benzodiazepines or gabapentinoids to treat pain and DO re-evaluate routinely for deprescribing during chronic use.
- DO NOT prescribe tramadol without consideration of the potential risks and harms related to serotonergic excess, seizures, falls, and drug-drug interactions (DDIs).
- DO NOT use strong CYP3A4 and P-glycoprotein inhibitors or inducers with DOACs and DO periodically assess for such DDIs.
The American Geriatrics Society (AGS) also worked on the CW campaign to identify treatments that potentially have more risks than benefits in older adults, concluding the following26:
- DO NOT use antipsychotics first-line to treat behavioral and psychological symptoms of dementia.
- DO NOT use benzodiazepines or sedative-hypnotics first-line for insomnia, agitation, or delirium.
- DO NOT use cholinesterase inhibitors for dementia without periodic assessment for perceived cognitive benefits and adverse gastrointestinal effects.
- DO NOT use antidiabetics to achieve hemoglobin A1c less than 7.5% in most older adults; moderate control is generally better.
- DO NOT use antimicrobials to treat bacteriuria in older adults unless they experience specific urinary tract symptoms.
- DO NOT maintain long-term proton pump inhibitor (PPI) therapy for gastrointestinal symptoms without an attempt to stop or reduce PPI at least once per year in most patients.
- DO NOT use NSAIDs for patients with hypertension, heart failure, or chronic kidney disease.
- DO NOT use prescription appetite stimulants or high-calorie supplements for treatment of anorexia or cachexia (unexplained wasting) in older adults.
- DO NOT prescribe medication without conducting a medication regimen review.
Prescribing Cascades
It bears repeating that providers should not initiate medications—prescription or over-the-counter (OTC)—or use a new medical device to treat new and emerging symptoms without first ascertaining that the new symptom is not an ADE of an already prescribed medication.24 This is the first CW statement endorsed by ASCP and it describes a prescribing cascade. Prescribing cascades are preventable, yet often unrecognized, even though researchers in the geriatric field first introduced the concept in the 1990s and then revisited and expanded upon it in 2017.27,28 They emphasized that “the identification and interruption of prescribing cascades is an important, actionable, and underappreciated opportunity to improve medication safety in older people.”27,28
Prescribing cascades contribute to polypharmacy and adverse outcomes in vulnerable older adults. By becoming familiar with the common prescribing cascades, outlined in Table 3, pharmacists and pharmacy technicians can identify culprit and cascade medications. If culprit medications remain clinically indicated, prescribers should use the lowest possible dose, a safer alternative with fewer adverse effects, or non-pharmacologic therapy, rather than initiating a cascade medication to address the ADE.
| Table 3. Prescribing Cascades to Avoid or Amend29 |
CULPRIT MEDICATION  |
ADVERSE DRUG EVENT  |
CASCADE MEDICATION |
| ACE inhibitors |
Cough |
Cough suppressants |
| Alpha-1 blockers |
Orthostatic hypotension |
Meclizine |
| Amiodarone |
Tremor |
Lithium |
| Hypothyroidism |
Thyroid hormones |
| Amitriptyline |
Cognitive impairment |
Cholinesterase inhibitors |
| Antipsychotics |
Cognitive impairment |
Cholinesterase inhibitors |
| Extrapyramidal symptoms |
Benztropine |
| Benzodiazepines |
Cognitive impairment |
Cholinesterase inhibitors |
| Beta blockers |
Depression |
Antidepressants |
| Bisphosphonates |
GERD |
PPIs/H2RAs |
| Bupropion |
Insomnia |
Sedatives-hypnotics |
| Calcium channel blockers |
Constipation |
Laxatives |
| Peripheral edema |
Diuretics |
| Cholinesterase inhibitors |
Insomnia |
Sedatives-hypnotics |
| Urinary incontinence |
OAB anticholinergics |
| Corticosteroids |
Insomnia |
Sedatives-hypnotics |
| Diuretics |
Urinary incontinence |
OAB anticholinergics |
| Dopaminergics |
Psychotic symptoms |
Antipsychotics |
| Gabapentinoids |
Peripheral edema |
Diuretics |
| Lithium |
Extrapyramidal symptoms |
Antiparkinsonians |
| Tremor |
Propranolol |
| Metoclopramide |
Extrapyramidal symptoms |
Antiparkinsonians |
| NSAIDs |
GI bleeding or heartburn |
PPIs/H2RAs |
| Hypertension |
Antihypertensives |
| OAB anticholinergics |
Cognitive impairment |
Cholinesterase inhibitors |
| SGLT2 inhibitors |
Urinary tract infections |
Antibiotics |
| SSRIs/SNRIs |
Insomnia |
Sedatives-hypnotics |
| Statins |
Myalgia |
NSAIDs/opioids |
| Thiazide diuretics |
Hyperuricemia or gout |
Allopurinol/colchicine |
ABBREVIATIONS: GERD = Gastroesophageal reflux disease; GI = Gastrointestinal; H2RAs = Histamine-2 receptor antagonists; NSAIDs = Non-steroidal anti-inflammatory drugs; OAB = Overactive bladder; PPIs = Proton pump inhibitors; SGLT2 = Sodium glucose cotransporter-2; SNRIs = Serotonin norepinephrine reuptake inhibitors; SSRIs = Selective serotonin reuptake inhibitors
Anticholinergic Medications
Older adults are more sensitive to anticholinergic adverse effects, including confusion, dry mouth, blurry vision, constipation, and urinary retention. A large case-control observational study of 58,769 patients diagnosed with dementia and 225,574 matched controls found statistically significant associations of dementia risk with exposure to anticholinergic medications, such as antidepressants, antipsychotics, antiparkinsonians, antiepileptics, and bladder anticholinergics.30 Participants who took a single strong anticholinergic medication daily for three years had almost 50% increased odds of dementia within a 10-year period. Dementia risk was increased in patients taking more than one anticholinergic medication due to the cumulative impact.30 Pharmacists can use one or a combination of multiple validated expert anticholinergic scales to calculate aggregate anticholinergic scores. An anticholinergic burden score of three or higher is associated with increased cognitive impairment and mortality. The most frequently validated scale is the Anticholinergic Cognitive Burden (ACB) scale.31,32 An Anticholinergic Burden Calculator is available at no cost for use here: https://www.acbcalc.com/.
A small longitudinal study of 69 participants found those who took an anticholinergic medication in combination with the cholinesterase inhibitor donepezil over a two year period declined by seven points on the Mini-Mental State Examination as compared with a three point decline in those taking donepezil alone.33 Thus, concomitant therapy with anticholinergics may counteract efficacy of cholinesterase therapy, resulting in a more rapid cognitive and functional decline.
Revisiting the case, Ms. Polly is taking donepezil for Alzheimer’s disease. Her prescriber consults with the pharmacist to initiate memantine for her progressive dementia due to increasing confusion and declining function. Medication review identifies several medications with anticholinergic effects that may be worsening Ms. Polly’s cognitive function, including cetirizine, diphenhydramine, furosemide, oxybutynin, and paroxetine. The pharmacist recommends deprescribing anticholinergic medications for which risk outweighs benefit. With reducing aggregate anticholinergic burden, donepezil may be more effective. This avoids added polypharmacy by initiating memantine, effectively preventing a prescribing cascade. Following discontinuation of these medications, the pharmacy technician can follow up with Ms. Polly and her caregiver to assess if there has been improvement.
Resources are available to assess polypharmacy and identify PIMs in vulnerable, older adults. The following section provides an overview, applicability, and utility in practice for the most common clinical tools: Beers Criteria, STOPP/START Criteria, STOPPFall Criteria, STOPPFrail Criteria, and the Medication Appropriateness Index.
Beers Criteria
The late Mark Beers, MD and his colleagues developed the Beers Criteria in 1991 with a mission to identify medications for which potential harm outweighed expected benefit and that should be avoided in long-term facilities. Since then, several updates have expanded the criteria to apply to older adults aged 65 years and older in ambulatory, acute, and institutionalized care settings, except end-of-life and hospice. Upon Dr. Beers’s death, AGS assumed responsibility for maintaining the criteria.
The 2023 AGS Beers Criteria for Potentially Inappropriate Medication Use in Older Adults is a well-known, widely available clinical tool to support shared clinical decision-making. It is available for free in the Journal of the American Geriatrics Society.34 For ease of clinical practice application, the AGS provides a pocket card and an app, for a fee, available at https://geriatricscareonline.org/. To encourage engagement and conversation, AGS provides patient and caregiver educational resources that are available for free at https://www.healthinaging.org/.
The AGS Beers Criteria lists medications and medication classes that the society and its geriatric expert panel consider potentially inappropriate for use in older adults. The criteria are structured by category with each section including a clear rationale and recommendation:
- Medications considered potentially inappropriate
- Medications potentially inappropriate in patients with certain diseases or syndromes
- Medications to use with caution
- Potentially inappropriate drug-drug interactions
- Medications to avoid or adjust dosages based on renal function
- Medications with strong anticholinergic effects
Importantly, the identified medications are “potentially inappropriate” rather than “definitely inappropriate.” The key word is “potentially,” as the prescribing and deprescribing processes are to be patient centric. The AGS panel recognizes that harm is typically more pronounced in the “old-old” than in the “young-old” and in patients with complex multi-morbidity and frailty.34 The indicators are categorized based on whether they involve avoiding the initiation of a PIM or discontinuing one that is already prescribed. To apply the AGS Beers Criteria, prescribers and pharmacists must identify the PIMs and, when clinically appropriate, consider safer pharmacologic or nonpharmacologic alternatives.
STOPP/START Criteria
The Screening Tool of Older Person's Prescriptions (STOPP) and Screening Tool to Alert doctors to Right Treatment (START) criteria are additional resources for identifying potentially inappropriate prescribing in older adults. These aim to identify PIMs and potential prescribing omissions, respectively.35 The STOPP criteria are most helpful in the context of polypharmacy. The original version, published in 2008, included 65 STOPP criteria, which more than doubled in the third version, updated in 2023, with 133 STOPP criteria. These more expansive criteria assist pharmacists and pharmacy technicians in identifying and preventing more DDIs, drug-disease interactions, and potential ADEs during medication reviews.
The STOPP criteria is divided into sections by organ systems—including cardiovascular, coagulation, central nervous, renal, gastrointestinal, respiratory, musculoskeletal, urogenital, and endocrine—with separate sections for drugs that increase fall risk, analgesics, and antimuscarinic or anticholinergic drugs.35 The criteria also address the need to clarify an indication for each medication, labeling the following as PIMs:
- Any medication prescribed without a clinical indication
- Any medication prescribed beyond recommended duration
- Any duplicate medication class
Additional variations of the STOPP criteria help healthcare providers to identify PIMs in older adults with a high risk of falls (STOPPFall) and in those with limited life expectancy (STOPPFrail).36,37 STOPPFall identifies medication classes considered to be fall-risk-increasing-drugs (FRIDs). To further assist in shared-decision making of deprescribing, practical decision trees based on the STOPPFall guidance are available online (https://kik.amc.nl/falls/decision-tree/).36
While most prescribing tools are designed to identify and prevent PIMs in the general older population, STOPPFrail consists of 27 potentially inappropriate prescribing indicators to assist healthcare providers in deprescribing in frail older adults.37 Deprescribing in frail older adults may improve their quality of life through a reduction in adverse events, hospitalizations, and mortality. Medication review in this population should focus on deprescribing and symptom management, rather than preventive and intensive treatment. STOPPFrail is applicable in adults aged 65 or older who meet all the following criteria37:
- End-stage irreversible pathology
- Poor one year survival prognosis
- Severe functional or cognitive impairment or both
- Symptom control is priority rather than preventing disease progression
Example criteria include37
- Avoid antiplatelets for primary cardiovascular prevention due to lack of evidence
- Bisphosphonates are unlikely to be beneficial for short-term osteoporosis treatment
- Aim for antidiabetic monotherapy with less stringent glycemic control (i.e., hemoglobin A1c goal of less than 8%)
End-of-Life Care
Prescribers and pharmacists should re-evaluate medications and minimize the pill burden for patients receiving palliative care, particularly at end-of-life (i.e., life expectancy of less than three months). Prevention of long-term complications is no longer indicated.21 Rather, the goal is comfort care through symptom-specific medications (SSMs) to treat pain, dyspnea (shortness of breath), nausea, cognitive disturbances, anxiety, and depression. The number of SSMs increases while the number of medications intended to treat comorbidities decreases. Therefore, the definition of polypharmacy shifts for end-of-life care, focusing on inappropriate medication use.
The shared decision-making process must still consider patient and caregiver preferences. While some patients may prefer only symptomatic treatment without life-prolonging measures, other patients may feel neglected following discontinuation of certain long-term medications.21 A literature review of 67 articles established preliminary recommendations for long-term medication classes shown in Table 4.21
| Table 4. Deprescribing at End-of-Life21 |
| MEDICATION CLASS |
RECOMMENDED ACTION PLAN |
GDR |
| Anticoagulants |
Discontinue for deep vein thrombosis primary prevention |
No |
| Antihyperglycemics |
Reduced dosages to prevent hypoglycemia |
Variable |
| Antihypertensives |
Discontinue for hypertension |
Yes |
| Antimicrobials |
Variable: goal is symptom control |
No |
| Statins |
Discontinue |
No |
ABBREVIATIONS: GDR = gradual dose reduction
Medication Appropriateness Index
Unlike the previously mentioned clinical tools that are PIMs and medication class lists developed by expert consensus; the Medication Appropriateness Index (MAI) is patient centric. The MAI, developed more than 30 years ago, is a patient screening tool that measures potentially inappropriate prescribing in older adults. The 10 simple questions focus on indication, effectiveness, dosing, duration, and potential for interactions to ultimately rate medications as appropriate, marginally appropriate, or inappropriate.38,39 A MAI Calculator is available at no cost for use here: https://globalrph.com/medcalcs/medication-appropriateness-index-calculator/.
HOW TO SAFELY DEPRESCRIBE
Deprescribing Tools
Once polypharmacy and PIMs are identified, many clinical tools are available to analyze benefit versus risk and guide pharmacy teams on how to safely discontinue specific medications and medication classes.
Deprescribing.org
https://deprescribing.org/resources/
Canadian Medication Appropriateness and Deprescribing Network
https://www.deprescribingnetwork.ca/professionals
Deprescribing.org is a website developed by Canadian pharmacists that provides guidance, tools, and approaches for deprescribing.40 Included within these resources are evidence-based guidelines and easy to use algorithms to determine when and how to stop specific medication classes: antihyperglycemics, antipsychotics, benzodiazepines, cholinesterase inhibitors, memantine, and PPIs.40 The companion site, Canadian Medication Appropriateness and Deprescribing Network, provides additional guidance on commonly inappropriate medications for healthcare providers and patients, including a Deprescribing Educational Program and brochures.41
US Deprescribing Research Network https://deprescribingresearch.org/resources-2/resources-for-clinicians/
The US Deprescribing Research Network is a tool funded by the National Institute of Aging to develop and provide information on deprescribing for older adults.42 The network’s efforts center around four “cores” addressing the needs of different stakeholders (e.g., researchers, patients) and a series of working groups focusing on research and standardized outcome measures. Resources for healthcare providers promote education by providing guides for specific medication classes and reference the deprescribing tools described above.42
Primary Health Tasmania https://www.primaryhealthtas.com.au/resources/deprescribing-resources/
Primary Health Tasmania is a not-for-profit organization in Australia that provides a comprehensive guide to personalized deprescribing, including principles, assessment of benefit versus harm, and patient and healthcare provider perceptions. These in-depth guidelines recommend deprescribing strategies for an extensive list of common medication classes: allopurinol, anticholinergics, anticoagulants, antiepileptics, antihyperglycemics, antihypertensives, antiplatelets, antipsychotics, benzodiazepines, bisphosphonates, cholinesterase inhibitors, gabapentinoids, glaucoma ophthalmics, inhaled corticosteroids, long-acting nitrates, NSAIDs, opioids, PPIs, statins, and calcium and vitamin D.43
MedStopper https://medstopper.com/
MedStopper is an online clinical tool for polypharmacy that assists healthcare providers, patients, and caregivers in the shared decision-making process and prioritization of deprescribing.44 Upon entering a patient’s regimen, MedStopper arranges the medications from “more likely to stop” to “less likely to stop,” based on the criteria in Table 5. A team of clinicians and researchers affiliated with Canadian healthcare institutions, including the University of British Columbia and the Centre for Effective Practice developed this site and is grounded in evidence-based resources such as the Beers and STOPP/START Criteria. However, it remains in the Beta testing phase, so pharmacy teams should still use it with caution.44
| Table 5. Deprescribing Opportunities and Prioritization44 |
| Medication |
Conversation |
| Potential to improve symptoms |
Is the medication helping symptoms? |
| Potential to reduce the risk of future illness |
Does the effectiveness justify the potential adverse effects, inconvenience, and cost? |
| Likelihood to cause harm |
Are symptoms caused by medications? |
Using the Edmonton Frail Scale, healthcare providers can identify patients as frail to adjust MedStopper’s “may cause harm” ranking. The specific indication for each medication affects the ranking categories for “may improve symptoms” or “may reduce future illness.” Categories are based on the rating levels of an unhappy, neutral, or happy face, with a color-coded discontinuation priority. Medications and indications also include links for resources, such as calculators for risk/benefit assessment, NNT, Beers Criteria, or STOPP Criteria. Healthcare providers can reference the tapering suggestions and symptom monitoring, as appropriate.44
Go back and review Ms. Polly’s medication regimen in Table 1. Which medications may be inappropriate based on the numerous prescribing and deprescribing tools that are readily available? Figure 2 shows a snapshot of a suggested plan for deprescribing utilizing the MedStopper clinical tool.
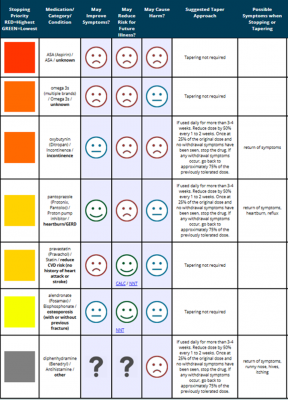
Figure 2. Deprescribing Plan Based on Stopping Priority by MedStopper
NOTE: Incomplete medication regimen
PAUSE AND PONDER: When do medications need a gradual dose reduction for discontinuation?
Pharmacist-Led Deprescribing
Research shows that comprehensive medication reviews may reduce PIM use and mortality in community-dwelling older adults aged 65 years and older.6 A narrative review of 25 global randomized controlled trials found that pharmacist-led deprescribing interventions had a beneficial effect on 69% of medication outcomes—including discontinuation, dose reductions, and medication changes—for patients with a mean age of 60 years or older residing in the community and nursing facilities.45
Pharmacists must follow a stepwise, patient-centric approach to deprescribing, while performing a comprehensive medication review of prescription and OTC medications as depicted in Figure 3. Since prescribing and deprescribing clinical tools do not consider preferences, life expectancy, and goals of care, it is essential to understand “what matters” to the patient and/or caregiver.
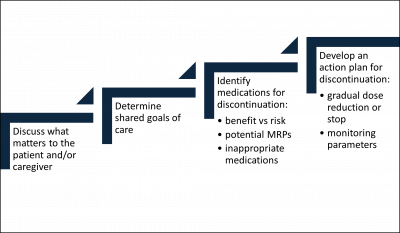
Figure 3. Stepwise Approach to Deprescribing
ABBREVIATIONS: MRPs = medication-related problems
Prescribers, pharmacists, and pharmacy technicians should assess if patients are not adhering to prescribed medications and, if not, why. Opportunities to deprescribe may be identified by asking just one question to a patient and/or caregiver. Pharmacy technicians are typically trained to ask, “do you have any questions for the pharmacist about your medication?” Instead, open-ended questions, such as “how do you feel about the number and types of medication you take?” or “how did your new medication help with your symptoms?” could gather more useful information. It is also important to ask if patients experience any adverse effects from their medications.
What Matters and Shared Goals
Deprescribing conversations ought to involve shared decision-making. Research shows older adults’ priorities are typically maintaining cognition, function, and independence over life extension.46,47 Patient preferences and health status change over time. Knowing a patient’s priorities and preferences about medication use helps align treatment decisions and guide deprescribing.
To determine a patient’s health priorities and preferences, healthcare providers can use the universal health Outcome Prioritization Tool (OPT) which is nonspecific to diseases and treatments. This simple tool elicits the preferences of older adults by having them consider trade-offs and rank the relative importance of four health outcomes46,47:
- extending life (regardless of health)
- preserving independence/maintaining function and activities of daily living
- reducing or eliminating pain
- reducing or eliminating symptoms (e.g., dizziness, fatigue, nausea, shortness of breath)
Several studies found that preserving independence and reducing pain were the most important health outcomes, whereas life extension was the least important.18,48,49 In the Older Patients’ Perceptions of Medicines and Willingness to Deprescribe survey, of the 50 participants, 76% wanted to keep their symptomatic medications and 61% wanted to keep their preventive medications.18
The “What Matters to You?” study of 350 patients used the OPT to assess the most important health outcome for older adults.50 Patients with cognitive impairment more often prioritized life extension. Importantly, cognitively impaired patients may struggle to understand health outcomes and the “trade-off principle”—prioritizing one health outcome while accepting potential deterioration in other health outcomes—which makes patients weigh and prioritize health goals. Patients with declining health status are generally more accepting of further decline. A secondary outcome found that the OPT is feasible for intensive treatment decision-making. For those patients who are unable to prioritize the four health outcomes, the OPT is still useful to facilitate a conversation about patient preferences and trade-offs to align decision-making.50
Considering Ms. Polly: how can the 4Ms aid in recommending oxybutynin discontinuation to her prescriber? “What matters to the patient is being able to continue to live in her home. Oxybutynin is anticholinergic and is negatively affecting her mentation and mobility as she has recent falls. Please consider a discontinuation trial of oxybutynin and non-pharmacologic options for overactive bladder.”
Identifying Medications for Discontinuation
Upon establishing goals of care, the next step is to engage the patient or caregiver and decide whether to deprescribe any medications. Common reasons for deprescribing include
- No active indication (e.g., health condition resolved)
- New or worsening condition
- Potential risks outweigh anticipated benefits
- Preventive indication with life-limiting conditions
- Lack of benefit or ineffectiveness
- Result of a prescribing cascade
- Nonadherence
- Interactions or ADEs
Prescribers and pharmacists can use the available prescribing and deprescribing clinical tools, but they must also review all medications and assess the benefit versus risk for individualized patients.
Developing an Action Plan
Upon identifying medications to deprescribe, prescribers and pharmacists must collaborate to develop an action plan to implement, monitor, and follow-up. When deprescribing, withdraw medications one-by-one by prioritizing those that are inappropriate and/or increase the risk of harm. It is critical to continuously consider “what matters” to the patient. Multiple medications can be withdrawn simultaneously if patients are experiencing ADEs, medications are easy to discontinue, or if they have minimal withdrawal effects.
If deprescribing is justified, prescribers, pharmacists, and pharmacy technicians need a plan of action for each medication (i.e., whether to abruptly stop or if gradual dose reduction (GDR) is necessary). Healthcare providers need to educate patients or caregivers on withdrawal symptoms to look for and emphasize the need to promptly report them.
Certain medications require a GDR or taper to prevent an adverse drug withdrawal event—meaning discontinuation results in clinically significant symptoms or signs—or worsened conditions.51 GDRs can identify the lowest effective dose, minimize symptom re-occurrence, and encourage patient willingness. For example, discontinuing benzodiazepines may cause agitation. If this withdrawal symptom is unfamiliar to healthcare providers, then a prescribing cascade may occur with initiation of an antipsychotic.43
Most medications have limited evidence and few guidelines for deprescribing. If no guidance is available, it is safer to attempt a GDR over weeks to months to discontinue rather than abruptly discontinuing. If a deprescribing guideline is unavailable, taper and discontinue medications in reverse of guideline recommended treatment. For example, the prescribing information for donepezil recommends initiation of therapy at 5 mg daily for four to six weeks, then increase the dose to 10 mg daily. To taper donepezil, decrease the 10 mg daily dose to 5 mg daily for four to six weeks and then discontinue therapy. Pharmacists can recommend a GDR plan in consideration of pharmacokinetics (e.g., half-lives), pharmacodynamics, dose, and duration of medications.51
To achieve successful deprescribing, a trusting relationship, ongoing communication, and adequate documentation is essential between prescribers, pharmacists, pharmacy technicians, patients, and caregivers. Using the word “trial” is reassuring to patients and caregivers since it implies patients can restart the medication if needed.
Returning to Ms. Polly, the pharmacist can initiate the conversation with her daughter by saying, “I understand you feel overwhelmed. It seems now would be a good time to take another look at your mother’s medications. We can ease the burden of her medication regimen and simplify it for her. This trial of stopping a couple of her medications may help with her confusion and lower her fall risk.”
CONCLUSION
Deprescribing is an essential part of good prescribing and an important strategy. It ensure older adults are not taking suboptimal medications. Effective and safe deprescribing requires a patient-centered, shared decision-making approach with evaluation of preferences, level of functioning, life expectancy, and goals of care. Numerous clinical resources are available to reduce polypharmacy, assess benefit versus risk, and provide guidance and approaches for how to discontinue specific medications. Deprescribing inappropriate or unnecessary medications impacts clinical outcomes—such as reduction in hospitalizations, ED visits, falls and overall healthcare costs—mitigates ADEs, and improves quality of life. Simplifying more complex medication regimens may also improve patient adherence to essential medications.
With a strong foundation in deprescribing principles and tools, it’s time to reevaluate Ms. Polly’s medication regimen one last time. What other medications should be considered for deprescribing to reduce polypharmacy, mitigate ADEs, and improve quality of life for both Ms. Polly and her daughter?